CASE REPORT
Ilio-mesenteric bypass for chronic mesenteric ischaemia where prior endovascular treatment has failed: a case series
Elsharkawy A,1 Khan K,1 Saleh F,1 Murray D,1 Richardson S1
Abstract
Background: Symptomatic chronic mesenteric ischaemia is a rare but debilitating disease, and its diagnosis is often delayed by the time taken to rule out underlying malignancy or other abdominal pathology. Once diagnosed, definitive revascularisation should not be delayed to avoid future bowel infarction. There is no consensus on the best treatment option, but most centres use angioplasty as the first choice, leaving open surgery for those who are unsuitable for or failed endovascular treatment. Failure of endovascular treatment does not seem to preclude open surgical bypass.
Methods: This series includes four patients who presented with symptomatic chronic mesenteric ischaemia in whom endovascular treatment failed and who were then managed successfully with ilio-mesenteric bypass, with average follow-up of 4 years.
Results: The average age of the patients was 57 years; three of the four patients were female. Two patients had initial successful angioplasty but required bypass later for recurrent symptoms. In the other two cases the endovascular approach failed immediately, with one developing acute ischaemia requiring bowel resection followed by mesenteric bypass.
Conclusion: Mesenteric bypass for symptomatic chronic mesenteric ischaemia is feasible after failed angioplasty. Immediate or delayed failure of endovascular treatment does not seem to preclude future surgery.
Introduction
Symptomatic chronic mesenteric ischaemia (CMI) is a relatively rare condition with an incidence thought to be in the range of 2–3 per 100,000.1 It is characterised by substantial morbidity, mortality and a decreased quality of life.2 The typical presentation includes postprandial abdominal pain, weight loss and/or unexplained diarrhoea. Mesenteric duplex ultrasound is an effective screening tool for mesenteric artery occlusive disease,3 while CT angiography is most often used for mapping the disease prior to any intervention, although magnetic resonance imaging is an alternative.4
Treatment targets for CMI are aimed at improving quality of life, restoration of normal weight and avoiding bowel infarction. The endovascular treatment of CMI has largely replaced open surgical management over the last two decades. The appropriateness of this shift has in part been validated by the findings of a recent meta-analysis.5 Open surgical revascularisation (OSR) was found to result in significantly more in-hospital complications and a trend towards a higher 30-day mortality compared with endovascular revascularisation (ER).5 However, these findings were balanced by superior long-term outcomes with primary and secondary patency rates being significantly higher for OSR according to further review articles.6,7
Current guidelines conclude that the reduction in short-term mortality and morbidity of ER outweigh the superior long-term results of OSR, particularly in view of the older population affected (mean age 69 years).8 Most centres, including our own, offer endovascular treatment as the first approach, leaving OSR for those who are not ER candidates or have failed ER. In this series we present two patients where initial ER was unsuccessful requiring OSR, and another two patients where previously successful ER failed, requiring OSR.
Methods
This was a retrospective review of patients who underwent intervention for CMI at Manchester Vascular Centre over the last decade. Those who had primary surgical bypass were excluded. From about 46 patients who had endovascular treatment, four patients who had failed angioplasty and required surgical bypass were included in this report. Hospital records for patients’ demographic characteristics, presentation, co-morbidities, intervention and re-intervention, outcomes and follow-up were analysed (Table 1).
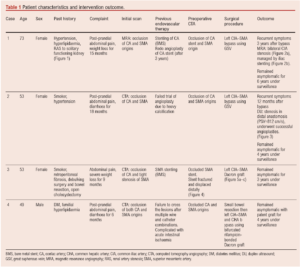
All patients underwent left common iliac artery (CIA) to superior mesenteric artery (SMA) bypass through a midline laparotomy incision. After inspection of bowel viability, exposure of the SMA was done by a longitudinal incision over the root of the mesentery facilitated by retraction of the transverse colon cranially and the small bowel medially. The left CIA was exposed by division of the retroperitoneum lateral to the sigmoid, avoiding injury of the ureter as it crosses the CIA. After administration of 5000 IU heparin intravenously, the inflow and outflow arteries were clamped sequentially and the graft was tunneled retroperitoneally in a wide C-shaped configuration to avoid kinking. Afterwards, the proximal anastomosis to the CIA was constructed first, followed by the distal one to the SMA. We used the greater saphenous vein (GSV) as a conduit in cases 1 and 2 (Table 1) and a prosthetic graft in the other two cases.
Postoperatively, all patients were prescribed antiplatelet and statin medications and were kept under surveillance protocol of combined clinical and ultrasound surveillance at 6 weeks, 3 and 6 months postoperatively, then every 6 months for 2 years and annually thereafter. The follow-up periods for our cases were 7, 4, 2 and 3 years, respectively.
Informed consent was obtained from all patients after explaining to them the nature and benefit of the study and thereby authorised reproduction of anonymised images.
Results
Four patients with an average age of 57 years (three women and one man) were included. All patients had occlusion of both the SMA and CIA origins on the initial scan (Table 1). In the first group (two patients) initial angioplasty was successful but required open bypass after 3 years from first angioplasty in case 1 and after 4 years in case 3 for recurrent symptoms. In the second group (two patients) initial trial of angioplasty failed. One patient was treated electively with OSR and the other patient suffered acute on chronic ischaemia with bowel infarction as a result. This necessitated exploration, bowel resection then OSR. There was no record of any major complications or mortality after OSR, and all bypasses remained patent for 8, 5, 3 and 4 years, respectively, until the time of writing this paper.
Discussion
In symptomatic patients with CMI, a conservative approach has no role in treatment and delayed revascularisation has been associated with clinical deterioration and bowel necrosis.9 Although there are no randomised controlled trials comparing the different treatment modalities for CMI, most centres advocate mesenteric angioplasty as the first option given the possibility of less perioperative morbidity and mortality.8
The results from a recently published large cohort of 245 cases from Denmark showed that endovascular treatment as the first option for CMI carries a 3-year mortality rate of 25% and a low risk of symptom recurrence.10 A meta-analysis by Alahdab et al5 comparing mesenteric ER with OSR, which included 100 observational studies and 18,726 patients, showed that OSR was associated with a statistically significant increased risk of in-hospital complications and a trend towards a higher 30-day mortality. In addition, the ‘endovascular first’ approach may lengthen the total successful revascularisation duration. One of our patients benefitted from a total of 4 years of primary and secondary patency of CA stent before having her bypass. There is evidence in the literature of more complications and a higher mortality rate in patients who required surgical bailout after failed ER,11 which raises the question whether an endo-first approach is appropriate for allcomers. Further research is needed to clarify this. However, in our series failed ER either immediately or late did not preclude successful OSR.
These early benefits of ER should be balanced against the superior long-term outcomes of OSR. Our series reports excellent patency rates for OSR with one bypass remaining patent for more than 8 years. This is consistent with meta-analyses comparing OSR and ER for CMI. In a systematic review of 1,795 patients by Pecoraro et al,6 the open surgical group has superior primary and secondary patencies. Also, Gupta et al7 concluded that the 5-year primary and assisted primary patencies were significantly higher in the OSR group, as well as freedom from symptoms at 5 years which was 4.4 times higher in the OSR group compared with the ER group.
We could argue that, in the setting of the ‘endovascular-first’ era, surgeons are performing fewer open mesenteric bypasses probably due to subspecialisation and centralisation of the service and the need for two surgeons to be present for open surgery. That might lead to a selection bias in the multidisciplinary team meetings for ER in patients whose disease pattern may be better served by OSR as the first approach. These patients would benefit from the longer patency rates and freedom of symptom recurrence that OSR offers. In addition, ER attempts for unsuitable lesions could result in devastating complications. In one of our patients an ER attempt complicated by distal embolisation down the mesenteric arcades required emergency exploration and bowel resection.
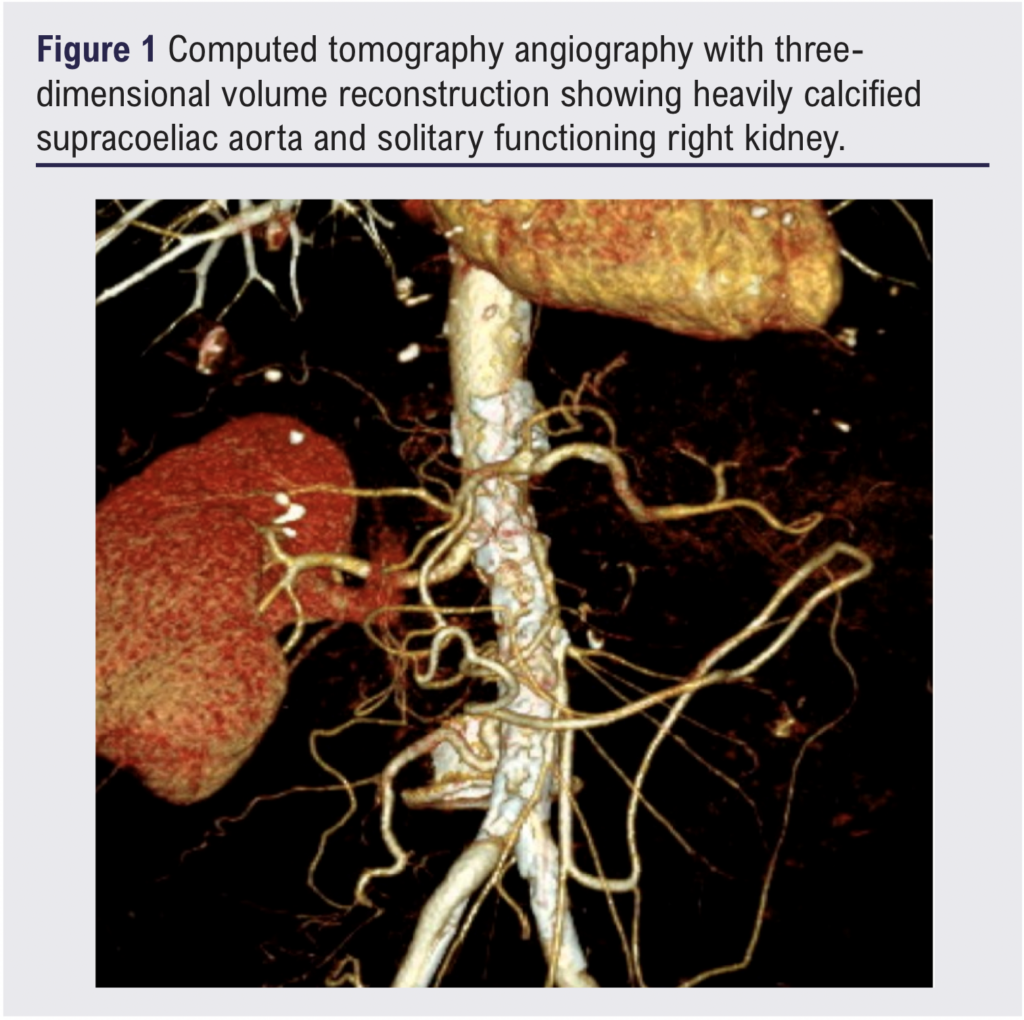
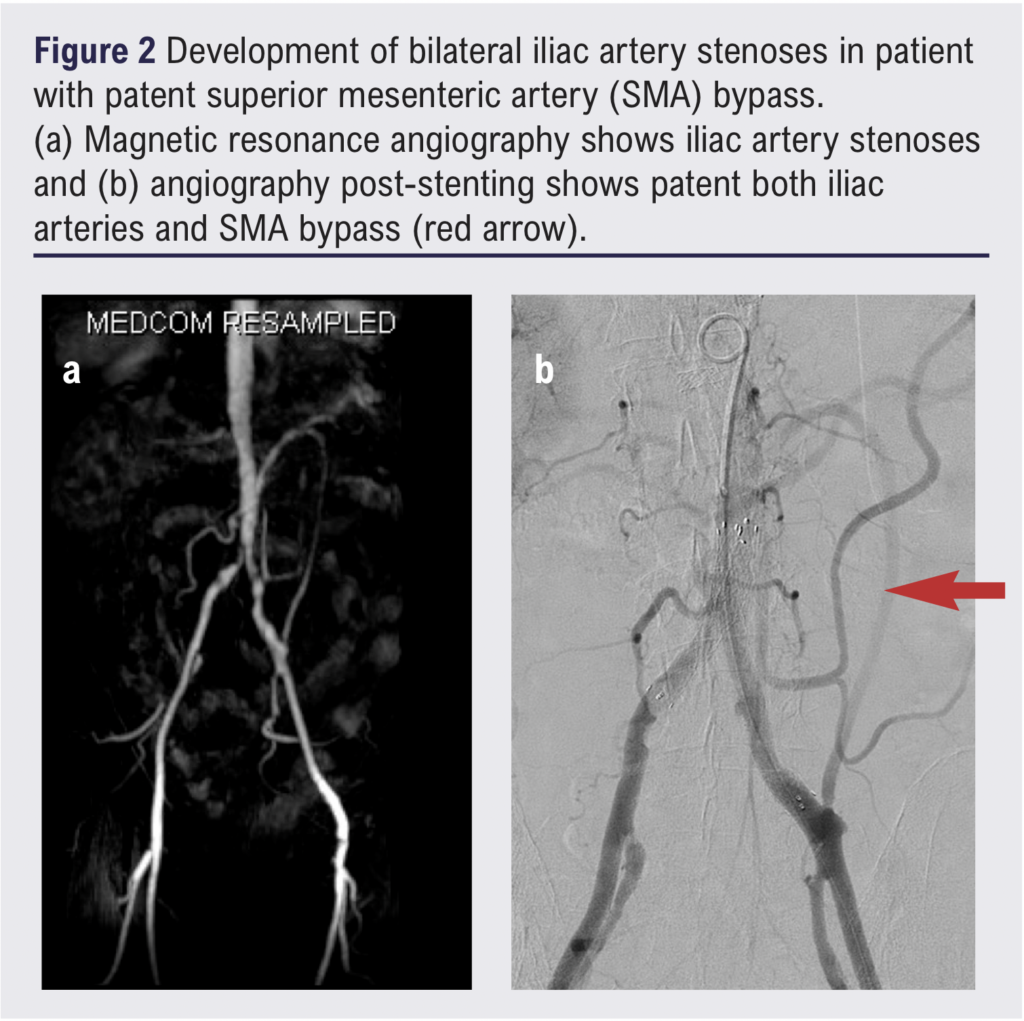
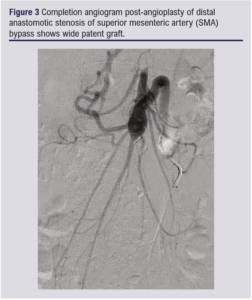
Secondary interventions to maintain the patency of mesenteric bypass, particularly when the GSV is used, are not uncommon. In the first case (Table 1) the patient developed recurrence of symptoms together with bilateral lower limb claudication 3 years after the bypass. Both duplex ultrasound and MRA confirmed development of bilateral CIA stenoses proximal to the mesenteric bypass which were treated successfully with bilateral iliac stenting (Figure 2a and b). Over the following 2 years, recurrence of symptoms confirmed with raised velocities on duplex ultrasound at the proximal anastomosis was managed with balloon angioplasty on two different occasions. In case 2 (Table 1), recurrent symptoms after 12 months of bypass and velocity of 812 cm/s detected by duplex ultrasound at the distal anastomosis were successfully treated with angioplasty (Figure 3). This emphasises the importance of close clinical and duplex ultrasound surveillance for these patients.
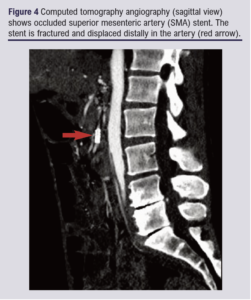
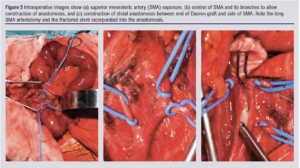
Conclusion
The series presented shows that OSR for symptomatic CMI is safe and effective following failed endovascular treatment. All patients may require strict clinical and radiological surveillance to ensure patency of the bypass.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2022;1(4):141-145
Publication date:
June 28, 2022
Author Affiliations:
1.Manchester Vascular Centre, Manchester Royal Infirmary Hospital, Manchester, UK
Corresponding author:
Mr Ashraf Elsharkawy
Manchester Vascular Centre, Manchester Royal Infirmary Hospital, Oxford Road, Manchester M13 9WL, UK
Email: ashraf.elsharkawy@ mft.nhs.uk











