ORIGINAL RESEARCH
The impact of the COronaVIrus Disease 2019 (COVID-19) pandemic on the clinical management of patients with vascular diseases: findings from Tier 3 of the COVID-19 Vascular sERvice (COVER) study
The Vascular and Endovascular Research Network COVER Study Collaborative, The Vascular and Endovascular Research Network (VERN)*
Plain English Summary
Why we undertook the work: COVID-19 caused major disruption to the ability of hospitals to provide normal care for patients suffering from vascular conditions. The aim of this study was to evaluate how the pandemic affected the treatment delivered to patients with vascular conditions. It took place over a 4-week period during the first wave of the pandemic.
What we did: Vascular surgeons in 19 countries took part, collecting information from 1,801 patients. For every patient referred urgently to the emergency on-call vascular team during the period, the treatment provided was recorded and if/how that differed from the ideal treatment plan. Treatment differences were graded from 1 to 5 to illustrate how serious the changes were. Minor changes, such as a different imaging technique used, scored 1. Major life-changing differences, such as amputation where otherwise a patient would have been treated with an operation to save their limb, were scored as 5.
What we found: The study found that globally as many as one in five people received a different treatment compared with what they would have had prior to the pandemic. This included one in three people with abdominal aortic aneurysm, one in five people with carotid stenosis and one in 10 people with diabetic foot disease. One-third of changes to care were classed as major, the most common being non-surgical treatment or delayed surgery for a patient who would normally have received immediate or urgent surgery. Very rarely, clinical care was improved such as the increased access to urgent hot clinics, resulting in shorter waiting time and admission avoidance, for those with diabetic foot infections.
What this means: This study demonstrates that many patients did not receive ideal care for key vascular conditions during the first pandemic wave. We can see that the policy changes and updates to guidelines were rapidly implemented and that hospitals were responsive to these changes. Interestingly, as many as one in five of the changes from ideal care were related to patients’ avoidance of hospitals for fear of catching coronavirus. If coronavirus-free surgical hubs are to be used going forward, patient perception of the safety of these centres will be key to their success.
Abstract
Background: During the first wave of coronavirus disease-19 (COVID-19), the pressure on healthcare led to significant restrictions and rationing of available services. Globally, vascular services were forced to change clinical management for a range of common, acute vascular presentations. The COVID-19 Vascular sERvice (COVER) Study Tier 3 aimed to understand the severity and impact of those changes on vascular patient referrals and acute presentations as a result of the pandemic in a global vascular cohort.
Methods: A 4-week multicentre multinational prospective observational study was launched in March 2020. Any hospital accepting urgent on-call vascular referrals during the pandemic was eligible to participate. Clinicians were asked to outline their actual management plan for each individual patient and to identify if the COVID-19 pandemic had changed their decision making. If so, clinicians then outlined their ‘ideal’ (pre-pandemic) management plan. All cases where management plans differed from that of the ‘ideal’ pre-pandemic management underwent further analysis to quantify the severity of the alteration on a scale of 1–5 (1: minor deviation in management, including imaging modality change, 5: palliation or amputation, where otherwise a patient would have been treated).
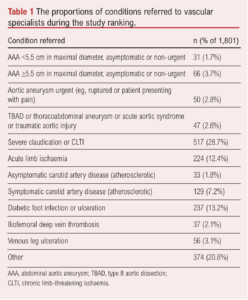
Results: A total of 1,801 patient episodes from 52 centres in 19 countries were included. The most commonly referred vascular conditions were chronic limb-threatening ischaemia (n=517, 28.7%), diabetic foot infections (n=237, 13.2%) and acute limb ischaemia (n=224, 12.4%). Only 2.3% of patients had a confirmed diagnosis of COVID-19. Deviations in management from pre-pandemic treatment occurred in 34.8% of those with unruptured >5.5 cm diameter abdominal aortic aneurysms, 27.0% of people with symptomatic carotid artery disease, 17% of people with acute or chronic limb-threatening ischaemia and 12.7% of people with diabetic foot conditions. Of these modifications, 40.7% were categorised as significant (grade 3a/3b) and 38.1% as major (grade 4), such as non-operative instead of operative management of carotid and lower limb disease. Life-changing/ending plans including major amputation or palliation, where pre-pandemic patients would have been offered limb or life salvage procedures, were made in 4.9% of cases (grade 5).
Lessons learnt: The results of this study suggest that the clinical disruption experienced by vascular surgery patients during the COVID-19 pandemic was largely due to redistribution of resources rather than individual patient infection. Guidelines for service modifications were adhered to, leading to significant changes in clinical management. One in five episodes of change in management were due to patient avoidance of clinical areas for fear of contracting coronavirus. If surgical hubs, with a low risk of COVID-19 infection, are to be utilised, then patient perception of the safety of these centres will be key to their success.
Conclusion: The COVER Study Tier 3 was able to show that close to one in five clinical vascular management plans were affected globally during the pandemic, with a small proportion leading to life-changing outcomes.
Introduction
The COronaVIrus 2019 (COVID-19) pandemic resulted in major changes in medical practice and clinical decision-making. Throughout the pandemic, the decision of whether, and how, to offer medical or surgical treatments was influenced by several factors such as COVID-19 positivity, patients’ risk of contracting COVID-19 whilst receiving treatment, resource rationing, staff shortages and government guidelines.1,2
These considerations were pertinent for vascular patients as they typically have multiple comorbidities. Furthermore, vascular procedures are usually offered in tertiary centres, which were also hubs for managing COVID-19 patients, and suffered significant pressures on available resources.
To support vascular surgeons with decision making in resource-scarce settings, the international vascular community produced guidance for clinicians on how to manage patients during the height of the pandemic. One example was the Vascular Society of Great Britain and Ireland (VSGBI) COVID-19 guidance, produced in early 2020, recommending groups for delayed or conservative intervention during periods of greatest resource scarcity (presentations who would have received prompt and guideline-recommended surgery prior to the pandemic).3
The impact of COVID-19 across countries or regions differed greatly in terms of both the timing and the number of people affected. As a result, the scale of the changes to vascular patients’ treatment and decision-making during the COVID-19 pandemic remains unknown. The COVID-19 Vascular sERvice (COVER) Study was a prospective multinational observational study evaluating the impact of COVID-19 on vascular patients using qualitative and quantitative methodology across 53 countries.4 This report explores the findings from Tier 3 of the COVER Study. The aim was to examine decision-making for new referrals made to vascular units during the first wave of the pandemic and compare the care given with ‘ideal’ (or pre-pandemic) practice.
Methods
Study design
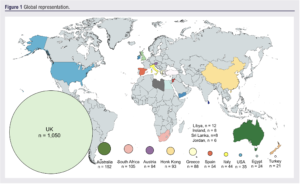
The COVER study was an international prospective cohort study (ISRCTN registration reference number: 80453162) designed and coordinated by the Vascular and Endovascular Research Network (VERN). The study protocol is available online and was published prior to commencing recruitment.4 Tier 3 of the COVER study presented here evaluated the type of care provided to patients who presented with a vascular pathology during the first wave of the pandemic. All referrals made to the emergency on-call vascular service were eligible patient episodes, even if the referred condition was subsequently deemed non-urgent. Any surgeon providing direct vascular care in a hospital setting was eligible to participate. Invitations were sent via email and social media in March 2020 by the VERN group. A total of 52 centres across 19 countries took part in the study.
Approvals
Ethical approval for the COVER Study was provided by a UK National Health Service (NHS) Research Ethics Committee (REC) and the UK Health Research Authority (HRA) (reference: 20/NW/0196 Liverpool Central). All regulatory approvals were granted prior to commencing patient recruitment in late March 2020. Non-UK centres obtained institutional review board approval (locally or regionally) according to their individual policies and in compliance with the Declaration of Helsinki.
Participants
Any patient with an emergency or urgent presentation referred to the participating institutions’ vascular services was eligible for inclusion, regardless of age or mode of referral. Participation in the study did not alter patient care in any way.
Data collection
Detailed records of how surgeons’ practice deviated from pre-pandemic care were completed on a per-patient basis. Electronic remote data collection proformas were designed, tested and refined by the VERN COVER team in March 2020. Baseline data for each patient were captured prospectively upon patient referral or presentation. This included demographics, type of condition or nature of referral, co-existing health conditions, medications, American Society of Anesthesiologists (ASA) physical status classification, frailty, as well as details relating to the presenting vascular complaint.
Clinicians were asked to report their current plan using a multiple options list specifically designed for each of the vascular conditions. This included information on whether treatment occurred as an inpatient or outpatient, whether patients underwent non-invasive or invasive imaging, and the nature of any endovascular or operative intervention(s). Clinicians were then asked whether their plan for each patient had been modified or impacted upon due to constraints specifically related to the pandemic. If so, they were also asked to detail their ‘ideal’ management plan as it would have been pre-pandemic, using the same list of multiple-choice options.
A vascular change in practice specific score was developed by the study team to permit reporting of the severity of the change in practice (see Supplementary figures at www.jvsgbi.com) for all cases with a reported change of management plan.
The VERN COVER team (12 vascular specialists from centres, UK-based) reviewed all possible iterations of management changes for each clinical condition and scored each change using the following grading system:
1. Minor deviation in management – eg, imaging modality change (provided recognised form of imaging); changing which oral medication is prescribed; changing the intravenous medication prescribed.
2. Minor deviation in management, which can have a more significant impact – eg, changing of medication from intravenous to oral; change in outpatient movement/flow, including seen in a different clinic; not being discussed in MDT; not having assessment of fitness (pulmonary function testing); not referring to specialist when otherwise would have.
3. Deviation from practice – eg, being managed as an outpatient when would have normally been inpatient; increase in threshold of intervention (for abdominal aortic aneurysm (AAA) surgery).
a. Less significant deviation in surgical/interventional practice or threshold – eg, ward-based amputation versus theatre-based amputation; slight technical difference in treatment (ie, different type of surgical bypass, different type of endovascular intervention).
b. More significant deviation – eg, endovascular intervention rather than open surgery for treatment of AAA.
4. Major deviation from practice – eg, amputation versus revascularisation; no revascularisation versus revascularisation; major amputation versus minor amputation.
5. Palliation or major amputation, where otherwise a patient would have been treated.
If there were multiple changes to a patient’s management pathway, they were allocated a score based on the single most significant and severe change.
Data management
De-identified data were transferred to a secure UK NHS server based at the University of Birmingham. Data sharing agreements with each participating centre were in place to ensure data protection. Each centre was required to record local identifiers on a secure General Data Protection Regulation (GDPR)-compliant database to allow longitudinal data capture and linkage, overseen by the Research and Development Department of the University Hospitals of Coventry and Warwickshire, Coventry, UK, who also acted as study sponsor. No identifiable patient data were recorded or exchanged.
Data handling and analysis
Each participating centre identified and entered consecutive patient data over a single 4-week period; the start of the four-week time period was selected at the discretion of each centre after all necessary approvals were in place. A data lock was applied on 9th September 2020. Data are reported as raw frequencies and percentages. Normally distributed data have been presented as mean ± standard deviation (SD) and non-normally distributed data are presented as median (range) values. Statistical analysis was performed using SPSS version 26.0 (SPSS, IBM, Chicago, Illinois, USA) and the language and statistical computing software R (V6) utilising additional software libraries. Independent variables were treated as nominal factors. Numerical data were treated as continuous data to maintain power. Primary and secondary outcomes were treated as dichotomous variables. Completeness of data was interrogated with a threshold of 90% for participants and 70% for dependent variables. Logistic regression was undertaken for the primary outcome. Univariate analysis was performed on all independent variables as a prerequisite for inclusion in the multivariate analysis, with a set threshold of p<0.1.
Results
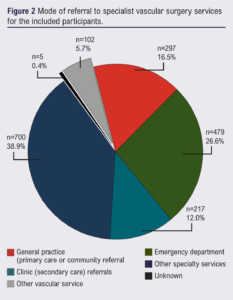
A total of 1,801 patient episodes were captured from 52 centres across 19 countries over a 4-week period (Figure 1). The largest proportions of referrals were made by speciality services such as secondary care physicians and podiatrists (Figure 2). Referrals from an emergency department constituted 26.6% of referrals.
The most common condition referred to vascular clinicians was chronic limb-threatening ischaemia (28.8%; Table 1).
A total of 41 patient episodes had a confirmed diagnosis of COVID-19 infection, 2.3% of the total 1801 episodes. Most patients were male (69%) and had diagnoses of hypertension and/or diabetes mellitus (Table 2). A pre-existing diagnosis of peripheral arterial disease was present in 31.5% of participants. Previous myocardial infarction was documented in 18.2% of patients, congestive cardiac failure in 7.8%, and/or angina in 5.5%.
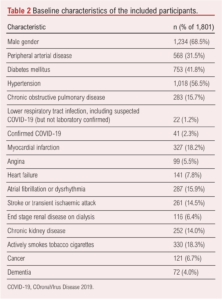
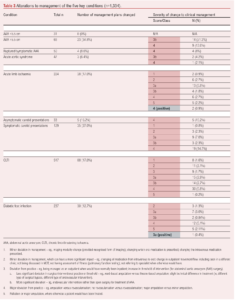
Table 3 shows a summary of the changes in management for each of the presenting vascular conditions. We present the specific findings for 1334 patients presenting with the following condition areas: AAA, acute aortic syndromes, acute limb ischaemia, carotid artery disease, chronic limb-threatening ischaemia and diabetic foot infection or ulceration (see Appendix 1 at www.jvsgbi.com for details of presentations across conditions).
Deviation from ‘ideal’ management
A total of 290 patient episodes (16.1%) had a confirmed change from the ‘ideal’ management plan. The highest proportion of patient episodes in which a change in management occurred was for those with AAA >5.5 cm in size (34.8%).
Regarding the severity of change in management, the proportion of patient episodes graded as 1, 2, 3a, 3b, 4 and 5 were 4.9%, 10.3%, 23.8%, 17.5%, 38.6% and 4.9%, respectively.
Aortic referrals
Overall, 30 patient episodes out of a total of 196 (15.3%) for those with aortic pathologies underwent a change in management. There were no changes in the management of the 31 sub-treatment threshold (<5.5 cm) AAA referrals. Of the 66 patients with an AAA measuring >5.5 cm, 34.8% (n=23) had a significant deviation in management; 14 (21.2%) scored 3, including a change from open to endovascular AAA repair (EVAR) despite suboptimal anatomy or a lack of cardiopulmonary exercise testing and anaesthetic review prior to treatment. Nine patients (13.6%) had a major deviation in management (score 4) which was predominantly an increase in the threshold to offer surgical treatment or delay to repair indefinitely. There were 50 ruptured or symptomatic AAA referrals, of which four (8%) had a major deviation (score 4 or 5) to their care compared with standard pre-pandemic practice: three (6%) underwent emergency open repair where EVAR would have been preferred but suitable theatre space or availability was limited, and in one case (2%) there was an issue with the endograft supply chain.
Forty-seven cases of acute aortic syndrome were referred but only three patients (6.4%) experienced a change to their planned management in the form of postponement of thoracic EVAR.
Acute limb ischaemia
A change in management occurred in 17% of acute limb ischaemia referrals (38 of 224 patients). Five patients (2.2%) with unsalvageable limbs received no intervention where, ideally, they would have undergone a major lower limb amputation (score 4). Six patients (2.7%) had a ‘major’ (score 4) change to their management which no longer involved an operative or interventional procedure (where ‘ideally’ indicated). The remaining changes were in the most part an alternative intervention or imaging modality, or a delay to intervention (score 2 and 3a).
Carotid artery disease
Overall, 40 patient episodes out of a total of 162 (24.7%) for those with carotid artery pathologies underwent a change in management. Major (level 4) changes to management were documented in 15.2% of patients, who were not offered a carotid endarterectomy (CEA) when they would have been pre-pandemic. There were 129 symptomatic carotid disease referrals with 35 (27%) having a documented change in management. Nineteen (14.7%) had a major change (score 4) and were offered best medical therapy only rather than either CEA or carotid artery stenting. Significant modifications (scores 3a and 3b) included delays to intervention beyond the recommended 14 days.
Chronic limb-threatening ischaemia
Of the 517 presentations, 88 (17.0%) received an alternative management plan to documented pre-pandemic ideal care. Half of these (45 of 88) had a significant or major adjustment (score 3a, 3b or 4), with one being palliated rather than being offered intervention (score 5). These significant or major deviations included delays to treatment and changing the operative modality to endovascular rather than open due to unavailability of endovascular suites or vice versa due to a lack of theatre space.
Diabetic foot infection or ulceration
Of the 237 patients seen with a diabetic foot infection or ulceration, 30 (12.7%) had a change in management. This included rare positive changes to care, such as access to urgent clinic assessment facilitated by a reduction in elective or other work, described in a single unit (positive 3a). For the remaining cases, the changes were adverse. Five cases (2.1%) had a major lower limb amputation (score 5) due to delays in presentation and access to services, which may have been avoidable if treated earlier. Twelve patients (5.1%) had wound and foot care only when ‘ideally’ revascularisation would have been indicated, constituting a ‘major’ modification (score 4). Ten patients underwent a ‘significant’ alteration (score 3a or 3b) to their plan with only ward-based foot care, toe amputation or sepsis drainage instead of operating theatre-based care under general or regional anaesthesia.
Drivers of change to management
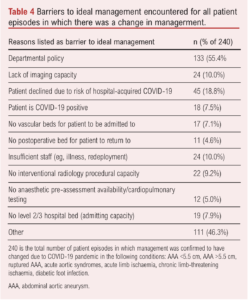
The clinicians’ reasons for having to provide management different from their ideal care were varied. A total of 240 patient episodes were confirmed to have a change in management across the chronic limb-threatening ischaemia, acute limb ischaemia, diabetic foot infection, carotid artery disease, AAA and acute aortic syndromes.
The barriers and stimuli for changes in management for these (grouped) conditions are listed in Table 4. The most common was department policy change for specific conditions (55.4%). The second most common reason was selected as “Other”, with 46.3% of patient episodes listing this. 18.8% of patient episodes with confirmed altered management listed “Patient declined due to risk of hospital-acquired COVID-19”. Other factors included a lack of operating space, lack of anaesthetic availability and a lack of suitable high-level care postoperative beds. Confirmed COVID-19 infection as a reason to change management was rare but did influence management negatively where indicated.
Discussion
Emerging single institution and surgeon surveys published during the first and subsequent waves of infection demonstrated significant variation in how vascular services managed the interruption to normal services, such as how surgery was rationed and the move from inpatient to outpatient management to reduce pressure on hospital beds.5,6 The COVER Tier 3 study has described these changes in more detail, and across a variety of countries and vascular centres.
As expected, lower limb emergencies comprised the most commonly referred condition, making up over half of the cases in this study (chronic limb-threatening ischaemia, acute limb ischaemia and diabetic foot infection/ulceration). The low proportion of emergency aortic pathology may reflect the global downward trend in AAA presentations, but does not take into account patients who died before reaching hospital or those who died at home while isolating.7 A change in referring practices from stroke teams in response to guidelines recommending medical management for all symptomatic carotids except for crescendo transient ischaemic attacks may have led to a lower proportion of those patients being picked up by this study.3
Uniquely, Tier 3 of the COVER study has attempted to quantify the effect of the COVID-19 pandemic on the management of individual vascular referrals. Data indicated that around one in three above threshold AAA and symptomatic carotid disease patients presenting during the pandemic were delayed. With AAA rupture rates increasing incrementally beyond 5.5 cm, and between one in six to one in 13 strokes thought to be prevented by carotid intervention for high-grade symptomatic stenosis, the potential loss of quality-adjusted life years could be high in the coming years.8 One in 10 chronic limb-threatening ischaemia presentations suffered delays and changes to management, and diabetic foot patients also suffered from delays to revascularisation and debridement. Guidelines have shown that non-optimised care in these groups leads to higher rates of major amputation and rising healthcare costs.9 This will place an additional burden on healthcare services globally.
The most common reason identified for a change in management was departmental policy, followed by “Other”. Pre-emptive action was taken in many countries to suspend elective activities and redeploy specialty surgical staff to intensive care and respiratory services prior to the peak incidence of COVID-19, reflecting the stimuli for change being departmental policy rather than individual patient-related or clinical factors. Selection of “Other” likely reflects the complex decision making, with multiple factors contributing to a change in management that cannot be put down as a restriction or lack of any given resource.
Patient perception of hospital-acquired COVID-19 was a barrier in as many as one in five patient episodes. If COVID-free surgical hubs are to be utilised effectively in the future, it will be important to address perception to ensure effective delivery of elective services.
As reflected in the prevalence of COVID-19 in this cohort, individual infection with COVID-19 was a barrier in as few as 7.5% of cases. This again reiterates that the vast majority of changes in management of vascular patients stemmed from a shift in service provision.
Limitations of the study
Whilst this study was designed with the intention of capturing all referrals to acute vascular services and to document the management plans accordingly, it is likely that there will have been a proportion of missed urgent cases, especially those in the community who never made it to the point of referral. The busy vascular take, coupled with workforce pressures and a shift to tele- or remote services,1,10 may mean that referrals received electronically or those triaged directly to outpatient clinics might not have been documented. Urgent or emergency vascular conditions, in general, carry a high associated mortality, in particular ruptured AAA or complicated aortic dissection. It is therefore possible that some cases may have been missed due to a lack of referral resulting from delays in presenting to medical services due to isolation or fear of catching COVID-19 in hospital. The timing for the 4-week data capture window was pragmatic and left to the participating institution to decide the start date. Consequently, in some cases this may not have aligned with the ‘true’ peak of the COVID-19 pandemic and could lead to under-representation of the true impact of clinical management changes. Long-term data out to 1 year from presentation was insufficient to provide information on impact on longer-term outcomes.
Furthermore, the scoring system used to grade the severity of change has not been validated. It was developed by UK-based vascular specialists and therefore may be biased towards UK-based practice.
Lessons learnt
The results of this study suggest that the clinical disruption experienced by vascular surgery patients during the COVID-19 pandemic was largely due to redistribution of resources rather than individual patient infection. The guidelines for service modifications, which were rapidly produced and published by the Vascular Society of Great Britain and Ireland, appear to have been taken up and adhered to both within the UK and further afield, leading to very real changes in clinical management during the first pandemic wave. Refinement of these guidelines should be a priority going forward, so that plans for service modification in the event of a future pandemic are available to expedite their timely publication.
It was interesting to note that one in five episodes of change in management were due to patient avoidance of clinical areas for fear of contracting coronavirus. If surgical hubs, with a low risk of COVID-19 infection, are to be used, then patient perception of the safety of these centres will therefore be key to their success.
Unfortunately, the lack of outcome data means it is difficult to interpret the impact of these changes in practice, and this is a limitation of the study.
Conclusion
Tier 3 of the COVER Study is unique in its description of management changes and the granularity of those changes. We have shown that decisions made for nearly one in five patients presenting during the pandemic were affected by significant or life-changing/ending alteration(s). Clearly these changes across the breadth of vascular surgery will lead to a surge of patients whose management was temporised in the initial pandemic wave.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2022;2(1):17-25
Publication date:
November 21, 2022
Author Affiliations:
* Full author information can be found in the footnote
Corresponding author:
Matthew Machin
NIHR Academic Clinical Fellow
in Vascular Surgery,
Department of Surgery & Cancer,
Imperial College London,
London, W6 8RF, UK
Email: [email protected].











