CASE REPORT
Use of a Heli-FX System Guide steerable sheath for internal iliac artery bridging stent re-intervention following iliac branch procedure via a contralateral femoral approach
Hossack M,1,2 Fisher R,1 Neequaye S1
Abstract
Endovascular aneurysm repair (EVAR) offers lower early morbidity and mortality at the expense of increased long-term re-intervention. Dilatation of distal landing zones is increasingly managed with iliac branch procedures. Re-intervention for endoleak from the internal iliac artery (IIA) bridging stent component is usually approached from arm access due to concerns regarding inducement of displacement of the EVAR caused by traversing the neobifurcation of the stentgraft from contralateral groin access. We present a novel case of successful re-intervention for IIA endoleak in the setting of previous fenestrated repair using the Heli-FX System Guide steerable sheath.
Introduction
Endovascular aneurysm repair (EVAR) is the dominant treatment modality for patients with anatomically suitable abdominal aortic aneurysms due to improved early morbidity and mortality over traditional open surgical repair. However, surveillance following EVAR is mandatory due to a higher incidence of late failure requiring secondary re-intervention.1 Dilatation of the iliac landing zones can lead to loss of seal and reperfusion of the aneurysm sac.2 Secondary interventions, which aim to extend the seal zones to non-diseased vessels, include embolisation and overstenting of the internal iliac artery (IIA) or preservation of IIA flow using an iliac branch procedure (IBP). IIA embolisation is associated with complications including buttock claudication (28% following unilateral embolisation, 42% following bilateral embolisation) and erectile dysfunction (19% and 24%, respectively). Bowel and spinal cord ischaemia can occur in 1–3% of bilateral embolisations.3 IBPs maintain IIA perfusion and thus reduce the incidence of these complications.4 Iliac branch device (IBD) implantation during EVAR is usually performed prior to aortic endograft insertion. Insertion of the iliac bridging stent can usually be achieved from contralateral groin access. It is generally accepted that insertion of the IIA component following previous EVAR requires upper limb access, as the traction exerted by a sheath from the contralateral groin over the neobifurcation of the stent graft can lead to endograft dislocation.5 However, brachial or axillary access is associated with a risk of arterial and peripheral nerve injury, and involves instrumentation around the aortic arch, with consequent risk of cerebral embolisation.6 Previous authors have reported a case of IBD implantation for re-intervention following EVAR using a steerable sheath from a contralateral femoral approach.7 We provide a novel report of re-intervention for failed IBD due to internal iliac component separation, using a steerable sheath from a contralateral femoral approach, in a patient with an existing EVAR with fenestrated extension.
Case report
A 75-year-old man underwent a planned re-intervention for internal iliac component separation resulting in a type IIIa endoleak following an IBP.
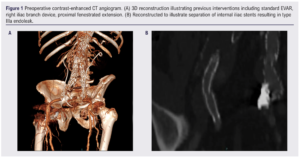
The patient had originally undergone standard EVAR (Talent; Medtronic CardioVascular, Santa Rosa, California, USA) for an infrarenal abdominal aortic aneurysm in 2008. In 2011, due to aneurysmal dilatation of the distal seal zones, he underwent a successful right iliac branch procedure (Jotec, Germany) and a left IIA embolisation with overstenting into the left external iliac artery (EIA). He was subsequently referred to our institution for a type 1a endoleak due to loss of proximal seal, and underwent a successful proximal extension with a 4-fenestration fenestrated cuff (Cook Medical, Bloomington, Indiana, USA) in 2018 (Figure 1A). Surveillance imaging revealed a type IIIa endoleak due to separation of the IIA bridging stent (9×57 E-Ventus BX, Jotec) (Figure 1B). The procedure was performed expeditiously due to reperfusion of the aneurysm sac by systemic arterial pressure. Arm access was considered but groin access was preferred due to the presence of stents to the visceral branches in the existing stentgraft lumen and increased risk of stroke with instrumentation around the aortic arch.
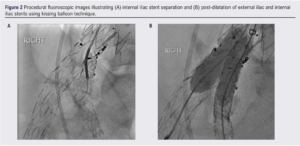
The procedure was performed in a hybrid operating theatre. Percutaneous contralateral femoral access was established using a standard preclose technique with two Proglide devices (Abbott Vascular, Santa Clara, California, USA). Initially a 12F steerable guiding sheath (Destino; Oscor, Palm Harvor, Florida, USA) was advanced from the left groin over the endograft flow divider. An 8F 90 cm flexor shuttle sheath (Cook Medical) was advanced co-axially and positioned above the origin of the IIA. The stent separation was crossed with a 0.035 hydrophilic glidewire (Terumo) (Figure 2A), but it was not possible to track any catheter or device across the lesion due to a lack of stability. The initial steerable sheath was exchanged for a 16F Heli-FX System Guide 22 mm steerable sheath which provided a more stable platform, allowing crossing of the lesion with a 4 mm angioplasty balloon, and pre-dilatation was performed. A 9×57 mm BeGraft Plus (BG+) (Bentley, Germany) over an 0.035 Amplatz Super Stiff 3 cm floppy tip (Boston Scientific, Marlborough, Massachusetts, USA) could not be tracked completely across the lesion, nor withdrawn due to stent slippage from the balloon, necessitating deployment with partial coverage of the lesion and proud of the IIA origin. This changed the geometry and facilitated crossing of the lesion with an 8×38 Atrium (Getinge, Sweden) to bridge the components and seal the type IIIa endoleak. The initial BG+ stent was seen to overhang and impinge on the flow into the EIA origin. Percutaneous retrograde right common femoral artery access was established with a 6F sheath. A self-expanding bare stent (12×60 E-Luminexx; Bard, New Providence, New Jersey, USA) was deployed in parallel. The EIA and IIA stents were post-dilated using a kissing technique to ensure equal expansion (Figure 2B).
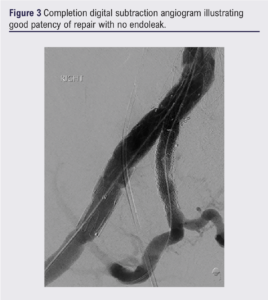
Final digital subtraction angiography revealed a good final result with patency of both branches and no endoleak (Figure 3). A 6F angioseal was used to close the right common femoral artery access. Left superficial femoral artery access was successfully closed using the preclose technique.
Discussion
EVAR has become the mainstay of treatment for anatomically suitable abdominal aortic aneurysms. Flared iliac limbs have been used to seal in ectatic or aneurysmal iliac arteries but subsequent degeneration is not infrequent and can result in distal failure.8 The European Collaborators on Stent/graft Techniques for aortic Aneurysm Repair (EUROSTAR) registry demonstrated an association between sealing in aneurysmal iliac vessels, aneurysm expansion and rupture.9 Thus, iliac re-interventions in patients with previous EVAR have been well described. Endovascular techniques for treatment of aneurysmal degeneration of the iliac landing zone include IIA embolisation and over-stenting into the EIA and IBPs, with IBPs having fewer complications. IBP following previous EVAR is complicated by the risk of damaging or dislodging the aortic stentgraft, which is thought to be higher when traversing the neobifurcation from a contralateral approach. Use of an upper limb approach for insertion of the IIA bridging stent has been described in efforts to minimise this risk.5,10,11 However, brachial or axillary access also carries a higher risk of cerebral embolisation and the vessels can be too small to approach percutaneously with the required sheath sizes. Several authors have described using contralateral groin access for IIA bridging stent insertion during IBP in patients with previous EVAR.7,12,13 Ferrer et al reported a case in which they successfully used a steerable sheath.7 Dawson et al described a novel ‘up-and-over’ technique using two sheaths ‘docked’ together, negating the need for a steerable sheath.13 Tadros et al reported two further cases in which this method was used successfully.12 The main advantages of this method is a cost saving and ability to use the manufacturer’s iliac bridging stent – therefore adhering to their instructions for use – which requires a larger internal sheath diameter than a balloon mounted covered stent.
Tenorio et al reported on the early and late outcomes of 53 patients treated with iliac branch endoprosthesis, including 13 patients with previous EVAR treated with the ‘up-and-over’ technique. The outcomes following the up-and-over technique for extension of previous EVAR were comparable to the standard technique used in de novo aneurysm repair.14
Regarding re-intervention following IBD implantation, Simonte et al reported on a 10-year single-centre experience including 157 consecutive IBD procedures.15 Freedom from IBD-related re-intervention was 97.4%, 95.6%, 94.0% and 91.8% at 1, 3, 5 and 9 years, respectively, whilst IIA patency was 94.7%, 92.6% and 90.4% at 1, 3 and 10 years, respectively. Owing to a more recent introduction of IBD technology and good long-term outcomes, there are few reports of re-intervention following IBD failure. To the best of our knowledge, this is this first report of re-intervention for IIA bridging stent separation from a contralateral femoral approach using a Heli-FX System Guide steerable sheath. By approaching from the contralateral groin, we avoided traversing the visceral segment of the aorta. However, use of a stearable sheath from the groin can also lead to inadvertent compression of visceral stents in the setting of previous fenestrated EVAR. Although the procedure was ultimately successful, it required changing of the steerable sheath. Interestingly, in this case the EVAR limbs were in a conventional configuration whilst, in other cases described,7,12 the limbs were crossed (so-called ballerina configuration), providing a wider curve over the flow divider. This may explain the instability we experienced with the initial 12F sheath.
Conclusion
Use of a Heli-FX System Guide 22 mm steerable sheath provides a more stable platform and facilitates a contralateral femoral approach for IIA bridging stent re-intervention, negating the need for arm access.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2023;2(2):114-117
Publication date:
January 30, 2023
Author Affiliations:
1. Liverpool Vascular and Endovascular Service, Royal Liverpool University Hospital, Liverpool, UK
2. Liverpool Centre for Cardiovascular Sciences, University of Liverpool, Liverpool, UK
Corresponding author:
Martin Hossack
Liverpool Vascular and Endovascular Service, Royal Liverpool University Hospital,
8C Link, Prescott Street, Liverpool L7 8XP, UK
Email: m.hossack@ liverpool.ac.uk











