ORIGINAL RESEARCH
WIfI scoring: a reliable tool for risk stratification in the diabetic foot clinic
Williams P,1 Bakewell Z,1 Akinlade B,1 Russell DA1,2
Plain English Summary
Why we undertook the work: Diabetes is estimated to affect over 5 million adults in the UK with a quarter developing a foot ulcer in their lifetime. 80% of people having their foot or leg amputated do so due to progressing diabetic foot ulcers. The associated cost to the NHS is around £1 billion per year. There are currently several scoring systems to help identify patients with the highest risk of amputation, the most commonly used being the Site, Ischaemia, Neuropathy, Bacterial infection and Depth (SINBAD) score. We compared a newer staging system (Wound, Ischaemia, foot Infection; WIfI) with the SINBAD to see if it more accurately predicts outcomes including amputation.
What we did: We reviewed the notes of patients who had been seen in our diabetic foot clinic over the 2 years from 2016 to 2018. People who had had a blood pressure measurement taken in the toe on the foot of their foot ulcer were eligible. We compared their eventual outcomes with their SINBAD score and their WIfI stage.
What we found: We found that WIfI stage 1–4 correlated better with time to healing, with stage 1 having the shortest time and stage 4 having the longest time. This was not the case with SINBAD severity. We found that a higher WIfI stage showed increased risk of foot/leg amputation at 1 year, whereas this did not change with SINBAD severity.
What this means: The WIfI stage more accurately predicted time to healing and gave a better idea of which patients will succumb to amputation at 1 year. This will help in several ways; it will allow patients to better understand their disease as well as providing a common language for future research to discuss different types of ulcers and their treatment.
Abstract
Background: The prevalence of diabetes is estimated to be over 5 million adults in the UK. Diabetic foot care is estimated to cost the NHS ~£1 billion per annum, meaning that diabetic foot ulcers (DFUs) are an increasing topic of discussion. It is estimated that reducing the prevalence of DFUs by a third could save the NHS up to £250 million annually. The Society of Vascular Surgery Wound, Ischaemia, foot Infection (WIfI) stage stratifies the risk of amputation and benefit of revascularisation in patients with threatened lower limbs and has been extensively validated in patients with chronic limb-threatening ischaemia, yet data on cohorts with diabetic foot ulcers in the UK remain scarce. The aim of this project was to compare the WIfI stage with the currently used Site, Ischaemia, Neuropathy, Bacterial infection and Depth (SINBAD) score in order to stratify risk in patients with DFUs.
Methods: The electronic case record (ECR) of eligible cases was reviewed retrospectively between February 2016 and March 2018. All patients with a recorded opening toe pressure were included. SINBAD score was taken from an ECR proforma and WIfI stage was calculated from ECR notes. The patients were followed up using electronic case notes.
Results: 119 patients with 129 foot wounds were included. WIfI stages predicted time to ulcer healing (p=0.04) whereas the trend for SINBAD severity did not reach significance (p=0.08). WIfI stages correlated with the proportion of patients with any minor/major ipsilateral lower limb amputation at 1 year (p=0.03) and minor amputation at 1 year (p=0.04), whilst SINBAD severity did not (p=0.95 and p=0.90, respectively). Conclusions: WIfI more accurately predicts time to healing than SINBAD severity. WIfI predicted amputation risk at 1 year but SINBAD did not. WIfI more accurately predicts risk of minor amputation.
Introduction
Diabetic foot ulcers (DFUs) are a common complication of diabetes, preceding over 80% of lower limb amputations in the UK.1 It is estimated that by 2025 more than 5 million people will have diabetes in the UK, with DFUs estimated to affect 25% of this population.1,2
There are several validated DFU classification systems that aim to provide risk stratification and enable comparisons of outcomes between groups of patients.3 Some systems address ischaemia to varying degrees as a contributing factor to amputations but are poor in reflecting other components that increase the risk of amputation.3,4 Currently, the SINBAD (Site, Ischaemia, Neuropathy, Bacterial infection and Depth) score is the most widely used in the UK and is a key component of the National Diabetes Foot Care Audit (NDFA), and has been well validated for wound healing and risk of amputation.3,5
The Society for Vascular Surgery Wound, Ischaemia, foot Infection (WIfI) classification system was designed to assist stratification of amputation risk and benefit from revascularisation in patients with a threatened lower extremity.4 The WIfI system uses wound, ischaemia and foot infection analogous to the tumour, node, metastasis (TNM) staging system in cancer and has since been established in several studies to correlate with wound healing time and amputation in patients with chronic limb-threatening ischaemia (CLTI),6-8 but its validity in cohorts of patients with DFUs is less well established.3,9,10
Background
Why are DFUs a problem?
Diabetic foot care is estimated to cost the NHS £1 billion per annum.11 Further research has revealed the mean cost of managing a single DFU over 12 months from initial presentation is £7,800, rising to £8,800 per unhealed DFU and £16,900 per amputated ulcer. DFUs significantly impact quality of life and survival. Data published by Diabetes UK suggest that four in 10 people who have a foot ulcer will die within 5 years and around half of those that experience a major amputation will die within 2 years.1 Improvements in wound care and lowering amputation rates significantly reduces the disease burden on the NHS. It is estimated that reducing the prevalence of DFUs by a third could save the NHS up to £250 million annually.12 This highlights clear clinical and economic benefits in improving outcomes.
Common DFU classification systems used in the UK include: The University of Texas Diabetic Foot Ulcer classification system (UT); PEDIS (Perfusion, Extent, Depth, Infection and Sensation) and SINBAD, the latter of which is the most widely used in the UK.3,4 More recently, WIfI has been introduced which may better quantify risk of amputation and also guide need for revascularisation. The SINBAD score is used in the NDFA in the UK and has been shown to correlate well with wound healing time in UK populations.5,13
The SINBAD score is calculated as shown in Table 1.
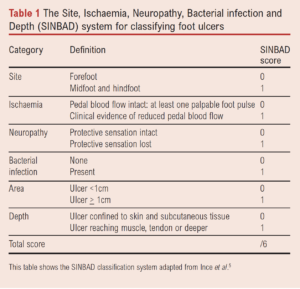 A score of >3 signifies a high-risk ulcer. In a worldwide population, risk stratification of ulcers is correlated with healing time.5 However, the SINBAD score does not give weight to the major factors affecting limb loss and revascularisation. SINBAD fails to account for the complex relationship between infection, ischaemia and wound healing.
A score of >3 signifies a high-risk ulcer. In a worldwide population, risk stratification of ulcers is correlated with healing time.5 However, the SINBAD score does not give weight to the major factors affecting limb loss and revascularisation. SINBAD fails to account for the complex relationship between infection, ischaemia and wound healing.
The WIfI scoring system was initially developed for CLTI, specifically in the diabetic patient.4,7 It aimed to move away from CLTI being defined by perfusion pressures alone.10 WIfI also aimed to treat the ischaemic aspect of foot ulcers as a continuum.10 It is calculated as shown in Table 2.
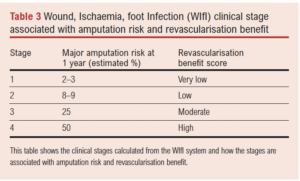
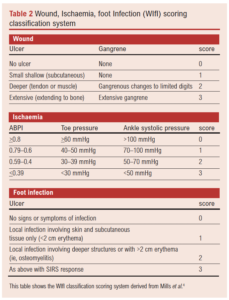 This then calculates a clinical stage which correlates to major amputation risk and revascularisation benefit as shown in Table 3.7-10
This then calculates a clinical stage which correlates to major amputation risk and revascularisation benefit as shown in Table 3.7-10
Given the limited data to validate WIfI in people with DFU, particularly in the UK, this project aimed to compare healing time, risk of amputation at 1 year and time free from amputation between both the SINBAD and WIfI systems.
Objectives
The objective of this study was to compare the SINBAD and WIfI classification systems in the risk stratification of DFUs. The primary outcomes were comparison of the proportion of patients undergoing amputation at 1 year between WIfI stages and high-risk and low-risk severity groups for the SINBAD score, and comparison of ulcer healing time between WIfI stages and SINBAD severity groups. The secondary outcome measure was the proportion of patients undergoing minor amputation between SINBAD severity groups and WIfI stages.
Methods
The study was performed in a tertiary referral diabetes limb salvage service (DLSS) at Leeds Teaching Hospitals NHS Trust. The study was registered and approved by the local Caldicott guardian. All patients with DFUs in the referral territory of the hospital are encouraged to be referred to the DLSS service for assessment and management. A DFU was defined as a break of the skin that involves the epidermis and part of the dermis below the level of the malleoli.14 At the time of the study, all patients were assessed with pulse palpation and audible handheld Doppler signal at first visit, along with 10G monofilament testing for neuropathy and ulcer assessment. Patients with palpable pulses or multiphasic handheld Doppler signals were deemed neuropathic and did not receive perfusion assessment. Further vascular assessment with toe pressures was only performed in the presence of peripheral artery disease detected with pulse and handheld Doppler assessment, or where the ulcer failed to reach a healing trajectory at 4 weeks. Toe pressures were performed using a commercially available device (Huntleigh, Dopplex ATP). The technique of toe pressure assessment has been well validated in other research so was not validated further in this study. Thus, the patients included in this study are ‘harder to heal’ than all comers attending the DLSS clinic. All ulcers were managed with ulcer bed debridement, dressings, offloading footwear and management of infection if present. Healing was defined as complete epithelialisation without discharge maintained15 as determined by the DLSS clinical team.
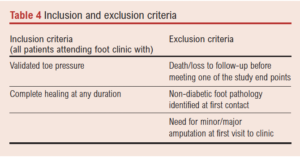
All patients with an opening toe pressure, diabetes and a foot ulcer for more than 4 weeks duration presenting between February 2016 and March 2018 were retrospectively analysed using electronic case notes. The inclusion and exclusion criteria are shown in Table 4.
 The SINBAD score was taken from the NDFA proforma of the case notes and the WIfI stage was calculated from data in the clinical records.
The SINBAD score was taken from the NDFA proforma of the case notes and the WIfI stage was calculated from data in the clinical records.
Statistics
The SPSS statistical package IBM SPSS Statistics for Windows, Version 25.0 (IBM Corp, Armonk, New York, USA) was used to perform all analyses. Differences in proportions were performed using a χ2 test, as were the comparisons for any amputation at 1 year and minor amputation. Due to the small number of major amputations, Fisher’s exact test was used to compare proportions. HbA1c was normally distributed and compared using one-way ANOVA. Time to event analyses were created using Kaplan–Meier survival curves with Cox regression. Time to event analyses followed patients up to 120 weeks for wound healing.
Risk stratification
In order to allow effective comparison between the two scoring systems and address the small number of patients within the higher number of groupings in the SINBAD score, we stratified the population into high severity and low severity groups based on their SINBAD score.5 Scores of 0–2 were defined as low severity and scores of 3–6 were defined as high severity. With respect to WIfI stage, patients were grouped based on their stage at presentation.
Results
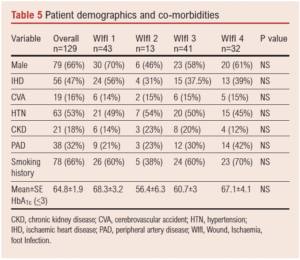
The study included 119 patients with 129 DFUs, with a mean age of 72±12 years and a male to female distribution of 66% vs 34%; 33% (n=43) of ulcers were WIfI stage 1, 10% (n=13) stage 2, 32% (n=41) stage 3 and 25% (n=32) stage 4 at presentation. The SINBAD score put 3% (n=4) in stage 0, 6% (n=8) in stage 1, 31% (n=40) in stage 2, 38% (n=50) in stage 3, 14% (n=18) in stage 4 and 7% (n=9) in stage 5.
The rates of ischaemic heart disease, cerebrovascular accident, hypertension, chronic kidney disease and HbA1c levels were evenly distributed between WIfI stages (Table 5).
Time to ulcer healing
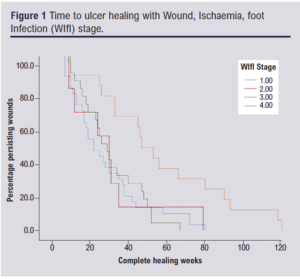
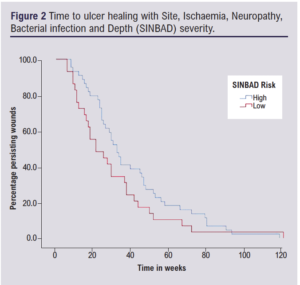
WIfI stage was associated with a significant increase in time to ulcer healing from stage 1 to stage 4 (p=0.04; Figure 1). Whilst a similar trend was seen with SINBAD severity, this did not reach significance (p=0.08; Figure 2).
Amputation at 1 year
There were 30 amputations during the 1-year period with seven being major, defined as any amputation at a level higher than the ankle. The remaining 23 were minor amputations. For the analysis on any (combined major or minor) amputation at 1 year, patients undergoing major and minor amputation on the same limb were considered as one amputation event.
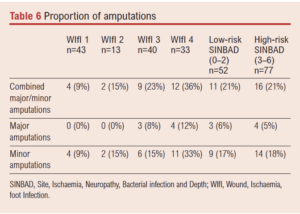
There was an increasing proportion of patients undergoing any amputation within 1 year with increasing WIfI stage (p=0.03) whilst there was no correlation with SINBAD (p=0.095). There was a trend towards an increased rate of major amputation with increased WIfI stage which failed to reach statistical significance (p=0.09), whilst the increase in proportion of patients undergoing minor amputation with increase in WIfI stage was significant (p=0.04). There was no clear differentiation of any category of amputation with change in SINBAD severity (Table 6; Figures 3 and 4).
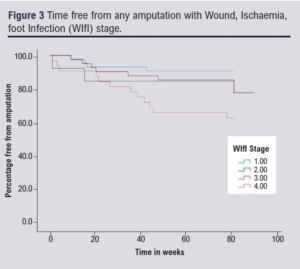
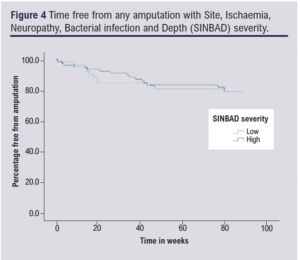
Discussion
The Society for Vascular Surgery WIfI stage has been shown to correlate well with healing time and amputation risk in a mixed cohort of patients with CLTI. It has previously been shown to correlate well with time to ulcer healing in a homogenous DFU population in the USA.9 Here we report the first UK-based data to show that WIfI stage both correlates with time to wound healing and predicts amputation at 1 year in a DFU population. This is in keeping with the wider literature when applied to more heterogeneous populations. The major amputation rate for our institution was 0% for both stage 1 and 2, 8% at stage 3 and 12% at stage 4. This is in contrast to the initial WIfI study in 2015 where a considerably wider variation in amputation rates was seen across WIfI scores (from 0% at stage 1 to 64% at stage 4).9 Earlier studies have found amputation rates up to 90% in stage 4 patients.7 Our findings are more in keeping with Robinson et al who studied a pure DFU cohort.8 The difference in results is likely due to a combination of two factors: differences in patient population and the ethos of the DLSS. Firstly, Zhan et al studied patients with CTLI which is a narrower and more severe spectrum of presentation than the data presented in this study. All of their patients were considered to have threatened limbs whilst our population included less severe disease. This would account for their higher rate of major amputation, especially in the highest risk groups. Second, the DLSS at our institution uses a multidisciplinary team approach to address not just the ulcer at hand but other systemic exacerbating or causative factors. Internationally this approach has been shown to reduce numbers of major amputations in DFU populations compared with care being delivered by vascular surgeons alone.9
Although we did not report a significant difference in major amputation rates between WIfI stages, there was a trend towards major amputation in the higher stages. Taking this in the context of the wider literature,8,9 this likely represents type 2 error given the small number of major amputations taking place in this cohort. We also report a significant correlation between WIfI score and minor amputation at any time (p=0.04). This is the first time this has been observed using the WIfI score in a DFU population, with the low-risk group having an 11% risk whilst the high-risk group had a 30% risk. For the purpose of this study, we defined minor amputation as any amputation below the ankle. These data could act as a baseline measurement for community teams trying to reduce the morbidity caused by DFUs in future studies.
We took the opportunity to compare the WIfI staging against the SINBAD score, which is currently the most widely used classification in the UK. Our findings would suggest that the WIfI stage is valid in a pure DFU cohort but, further to this, it is better at predicting outcomes than the SINBAD score in a cohort of patients with hard to heal DFUs in a specialist clinic. However, it should be recognised that 69% of ulcers in this study were evenly distributed between SINBAD scores 2 and 3, and the lack of a wide distribution of the ulcers across the range of SINBAD scores may in part account for the reduced differentiation in clinical outcomes associated with SINBAD severity in this study. The SINBAD score has proved to be a useful tool in evaluating DFUs, but where regular and consistent access to expert DLSS exists, the more objective measures required for WIfI should be undertaken to give better and more reliable prognostic information to patients and to better identify those patients who would benefit from early revascularisation.
There are limitations to this study. It is retrospective and therefore at risk of selection and observer bias. The study only includes those patients who had an objective measure of perfusion pressure and has therefore sub-selected a population who are likely to have harder to heal ulcers than the general population attending our clinic. Therefore, there needs to be some caution about the generalisability of these data, although it would be expected that the differentiation between low- and high-risk patients would be greater for both classification systems in a full patient cohort which would have a higher proportion of low-risk ulcers and better associated outcomes. There is inter-rater variability in WIfI and SINBAD in that the former was recorded retrospectively and the latter calculated prospectively by different clinicians using retrospective data. The absolute number of major amputations was small (n=7), which could affect the reliability of the measurements. However, our outcome is in keeping with the wider literature.9
Conclusion
This is the first study in a UK diabetic foot cohort to show that WIfI stage correlates with increased risk of amputation at 1 year. It also shows that WIfI better predicts time to healing than the SINBAD score. Despite the limitations of this study, it has shown the WIfI stage to be a valid tool for risk stratification in patients with a DFU in a UK population and should be widely adopted to the benefit of patients. The next stage in research would be a larger prospective cohort study examining outcomes and correlating them with the scoring system at presentation and eventual resolution of disease.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2022;1(3):71-76
Publication date:
April 25, 2022
Author Affiliations:
1 Leeds Vascular Institute, Leeds General infirmary, Leeds, UK
2 Leeds Institute of Clinical Trials Research, University of Leeds, Leeds, UK
Corresponding author:
Mr Paul Williams
Leeds Vascular Institute, Leeds General infirmary, Great George Street, Leeds LS1 3EX, UK
Email: [email protected]