ORIGINAL RESEARCH
Endovascular aneurysm repair using the Gore Excluder Conformable Abdominal Aortic Aneurysm Endoprosthesis with active control system in octogenarians with highly angulated aneurysm neck: a UK single-centre experience
Elsharkawy A, Mahmood A, Sirinivasamurthy D, Sayed S, Matharu N
Plain English Summary
Why we undertook the work: Endovascular aneurysm repair (EVAR) is a minimally invasive procedure to prevent an aortic aneurysm from bursting. It involves inserting a graft within the aneurysm through small groin incisions using X-rays to guide the graft into place. It is particularly useful in patients of advanced age with multiple medical problems who would be unsuitable for major open surgery. However, it requires certain anatomical features of aneurysm shape. Those with highly angulated aneurysm necks (the part of the aorta between the level of the kidney arteries and the sac) may represent a challenge for EVAR with most commercially available grafts. A new device manufactured by Gore medical company has been designed to accommodate this angulation and achieve a robust seal in this unfavourable anatomy. We think the use of this device in patients of advanced age with an unsuitable aneurysm shape is feasible with satisfactory early results.
What we did: We looked retrospectively at the hospital records of all patients aged >80 years who have been treated with this graft at our unit between January 2020 and July 2023. We assessed the outcomes, technical success, early complications and the rate of secondary intervention during the first 30 days after the procedure.
What we found: The initial technical success of this device is 95%, which means successful insertion of the device within the aneurysm in the absence of surgical conversion or death, graft blockage or significant leak of blood between the graft and aneurysm sac. None of the 20 patients included in the study who had this device implanted required secondary intervention during the first 30 days. No device-related deaths were reported in the same period.
What this means: This device enabled patients of advanced age with frailty and those with unfavourable aneurysm shape to be treated with EVAR according to the Instructions for Use designed by the manufacturer of the device. Although the initial results are encouraging, long-term outcomes need to be investigated further.
Abstract
Objective: Abdominal aortic aneurysm (AAA) with highly angulated neck (>60°) in octogenarians represents a challenge to treatment with endovascular aneurysm repair (EVAR). In such cases, EVAR may be used outside the manufacturer’s Instructions for Use and attendant higher risk of failure. The new device Gore Excluder Conformable AAA Endoprosthesis with active control (CEXC) system has been designed to achieve optimal aortic wall apposition and seal in these more challenging geometries. This study reports the initial results and technical success of this device in octogenarians with highly angulated aneurysm necks (>60°).
Methods: All patients aged >80 years who had AAAs treated with the CEXC device at a single UK centre between January 2020 and July 2023 were analysed. The primary endpoint was technical success, which was defined as successful introduction and deployment of the device in the absence of surgical conversion or mortality, type I or III endoleaks or graft limb obstruction. The secondary outcomes included 30-day morbidity, mortality, endoleak and re-intervention rates.
Results: Twenty patients were included (17 (75%) males) of median age 81 (range 80–84) years. The median AAA diameter was 60 (57–63) mm and the median infrarenal neck angulation was 80 (70–89)°. The active control system was used in six cases. The median procedural time was 277 (254–316) minutes. The immediate technical success rate was 95%, with one type Ia endoleak (5%). In the 30-day postoperative period, four complications were reported. On the 30-day follow-up CT scan, five type II endoleaks were observed and one initial type Ia endoleak had persisted at 30 days. No re-intervention was required during the first 30-day postoperative period. One hospital death occurred during the coronavirus disease 2019 pandemic; however, the death was unrelated to the surgery.
Conclusions: EVAR with the CEXC device in octogenarians with hostile aortic neck anatomy is feasible inside the Instructions for Use, and shows satisfactory initial results.
Introduction
In March 2020 the National Institute for Health and Care Excellence (NICE) guidelines recommended open repair as the primary treatment option for unruptured abdominal aortic aneurysms (AAAs), while suggesting endovascular aneurysm repair (EVAR) as a viable alternative for individuals with a high anaesthetic risk. Octogenarians constitute a subset of the high-risk population. They exhibit increased frailty and operative mortality rates of 5–10% in open repair procedures.1,2 Consequently, open repair is usually unsuitable for these patients, either due to the presence of multiple comorbidities or solely because of their advanced age. EVAR presents a less invasive alternative with reduced morbidity and mortality.3 This approach offers certain advantages, particularly in preventing decompensation in patients characterised by increased frailty but an otherwise favourable life expectancy. However, hostile neck anatomy continues to pose a challenge for EVAR. EVAR in severe neck angulation (>60°) has been associated with significantly higher rates of type Ia endoleak (EL), neck-related secondary intervention and device migration compared with those with non-severe neck angulation.4 The use of EVAR devices outside the manufacturer’s Instructions for Use (IFU) has been associated with reduced technical success rates and the increased use of adjunctive procedures.5,6 This is especially pertinent in the case of octogenarian patients with increased frailty, who would be expected to decompensate with multiple interventions. The newly introduced device, Gore Excluder Conformable AAA Endoprosthesis with active control (CEXC) system (W. L. Gore & Associates, Flagstaff, Arizona, USA) has been designed to address these issues.7 The active control system enables repositioning and adaptability of the device to the native aortic wall, thereby facilitating optimal wall apposition and aggressive infrarenal positioning. This, in turn, maximises the likelihood of achieving seal in complex aortic geometries and avoids early and late type 1aEL. Consequently, eligibility for EVAR within the IFU of this novel device has significantly expanded to include patients in their 80s with severely angulated aortic necks.
To date, only a few studies have explored EVAR with the CEXC device. Moreover, no reports exist on its use in octogenarian patients with increased frailty. This study aimed to present the first report of the immediate technical success and early outcomes associated with the use of the CEXC device in the treatment of highly angulated AAAs in octogenarian patients.
Methods
This study retrospectively reviewed data from 108 patients who underwent EVAR for AAAs at the University Hospitals of Coventry and Warwickshire (UHCW) between January 2020 and July 2023. The data for this analysis were obtained from the Department of Clinical Coding. Of the 108 patients, 20 individuals aged >80 years with severe neck angulation of AAAs (>60°) who were treated with the new bifurcated Gore Excluder CEXC system are included in this report. Patients who had EVARs with other devices were excluded from this study, as well as those with favourable anatomy, but the CEXC device used was based on the choice of the operating surgeon. A local study ethical approval has been obtained for this retrospective review.
Patient demographics, preoperative anatomical characteristics, procedural details, postoperative complications and follow-up details were reported. Baseline data, including age, sex, presence of hypertension, diabetes mellitus, ischaemic heart disease and chronic obstructive pulmonary disease, were collected from hospital records.
Anatomical characteristics of the aneurysms, along with all measurements, were recorded by a single observer from preoperative CT aortograms. The slice thickness of these CT scans was 0.625 mm. These measurements included the maximum AAA diameter, neck diameter, neck length and common iliac artery (CIA) diameters, all measured as the outer-to-outer wall diameter.
Operative details included technical success, utilisation of the active control system, the presence of ELs on completion angiography and the procedure time. Technical success was defined according to the Society of Vascular Surgery reporting standards as the successful introduction and deployment of the device in the absence of surgical conversion or mortality, type I or III ELs, or graft limb obstruction.8 As per our unit policy, all patients and their preoperative CT scans were discussed in multidisciplinary team (MDT) meetings involving multiple vascular consultants, intervention radiologists and vascular anaesthetists. After the deliberations, the patients were deemed unfit for open surgery. All procedures were performed by two consultant vascular surgeons, at least one with EVAR experience.
The 30-day postoperative complications, mortality, CT scan findings and re-interventions were recorded.
Statistical analysis
All data were analysed using IBM SPSS Statistics, version 29 (IBM Corp, Armonk, New York, USA). Histograms were inspected visually to assess normality. Non-normally distributed variables are presented as medians with interquartile ranges (quartiles 1–3) for continuous data and as numbers and percentages for categorical data.
Results
Demographic characteristics
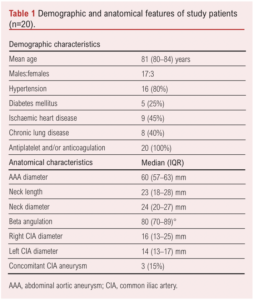
Of the 20 octogenarians treated with the CEXC device and included in this study, 17 (75%) were males and three (25%) were females. Sixteen (80%) patients were on antihypertensive medications while five (25%) were diabetic. Nine (45%) patients had ischaemic heart disease and eight (40%) had chronic obstructive pulmonary disease. All patients were receiving at least one form of antiplatelet therapy and/or anticoagulation, in addition to statins. Table 1 shows the demographic characteristics of the patients.
Anatomical characteristics
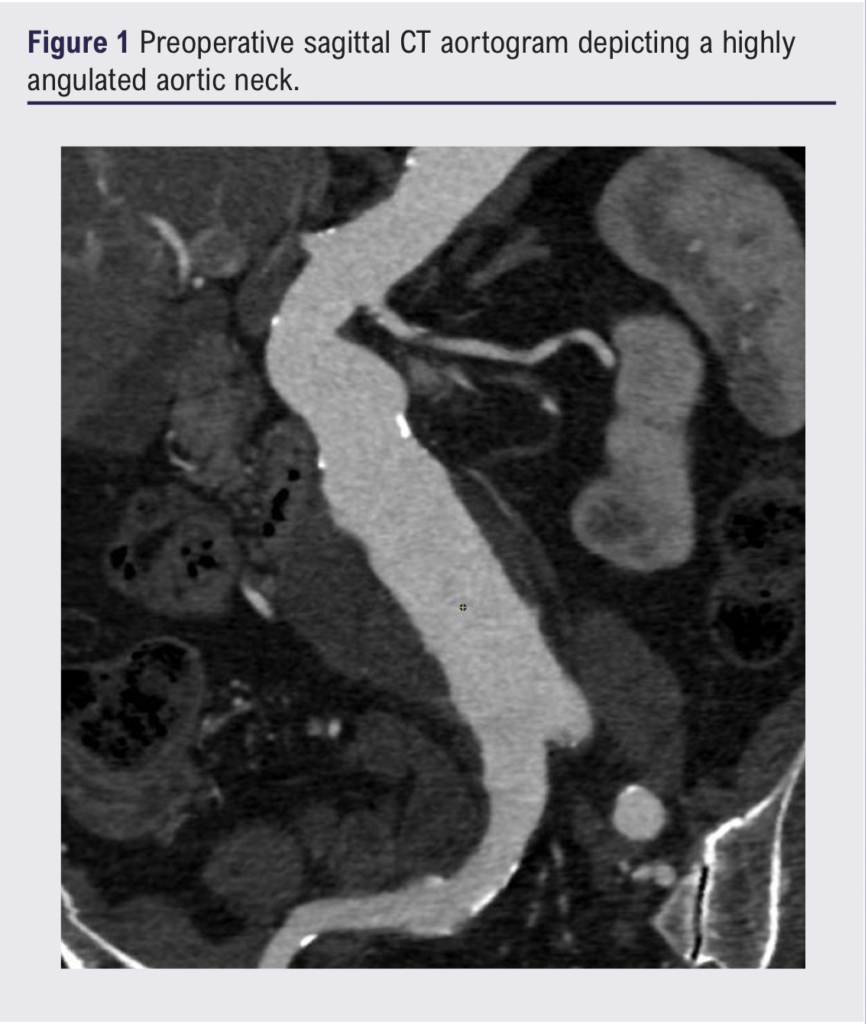
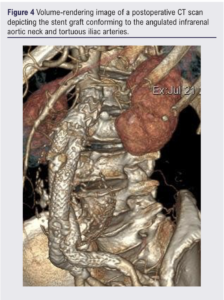
Table 1 summarises the preoperative anatomical features of the AAAs. The median AAA diameter was 60 (57–63) mm, neck length 23 (18–28) mm, neck diameter 24 (20–27) mm and infrarenal beta angulation 80 (70–89)°. The median right and left CIA diameters were 16 (13–25) mm and 14 (13–17) mm, respectively. CT imaging of severely angulated aortic neck and tortious iliac arteries is demonstrated in Figure 1.
Procedural characteristics
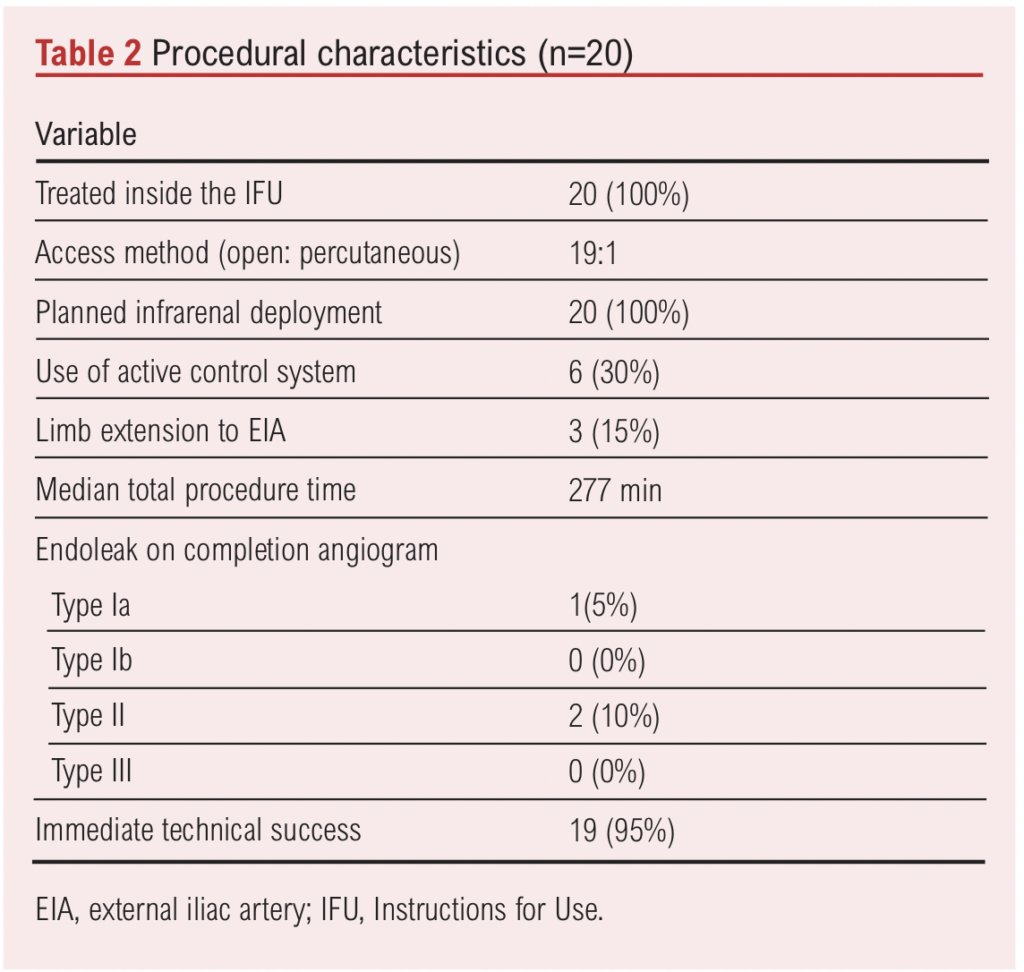
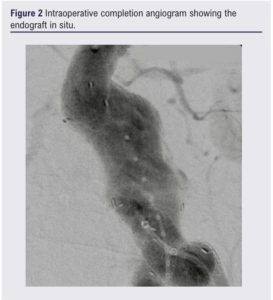
Table 2 shows the procedural characteristics. All patients (100%) were treated within the IFU of the device (neck length >15 mm and beta angulation >60°). The active control system was used in six (30%) patients to attain a proximal seal. In three (15%) patients, embolization of one internal iliac artery was required to enable limb extension to the external iliac artery for the treatment of a concomitant CIA aneurysm. The median procedural time was 277 (207–360) min. Technical success was achieved in 19 (95%) procedures, with one type Ia EL (5%). Additionally, two (10%) type II ELs were noted on intraoperative completion angiograms. However, these type II ELs were resolved on the 30-day CT scan. An intraoperative angiogram demonstrating the successful exclusion of the aneurysm with the endograft in situ is shown in Figure 2.
30-day morbidity and mortality
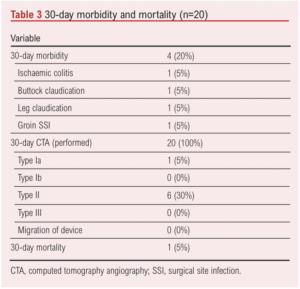
We observed four (20%) surgical complications: ischaemic colitis in one patient, right buttock claudication in one patient, common femoral artery occlusion in one patient and groin surgical site infection in one patient. All patients were managed conservatively and did not require any intervention.
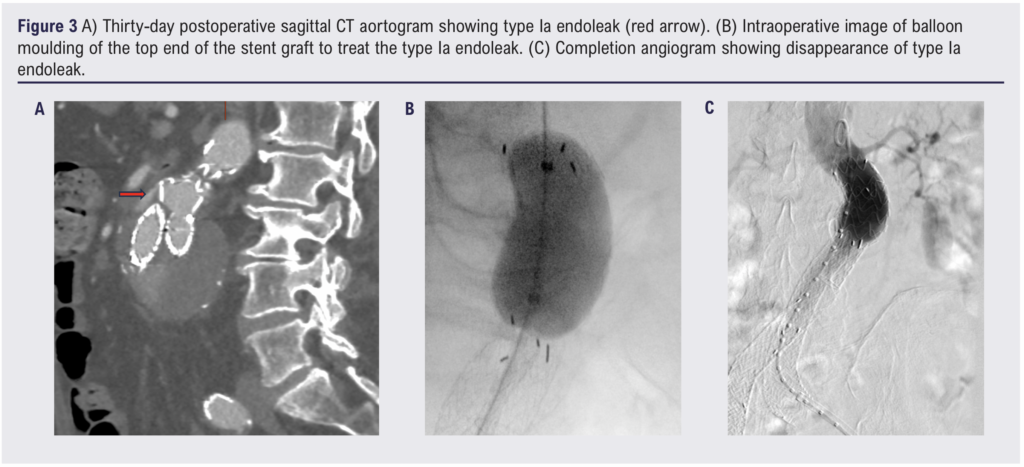
On the 30-day postoperative CT scan six (30%) type II ELs were observed; however, none of them required intervention. The initially observed type Ia EL on the completion angiogram has persisted on the 30-day CT scan. However, this has been successfully treated with balloon moulding of the top end of the graft, done under local anaesthetic on postoperative day 103 (Figure 3A–C). Figure 4 shows a 30-day postoperative CT scan demonstrating the Gore device conforming to the angulated aortic neck and tortuous iliac arteries. No re-intervention was required during the first 30-day postoperative period. One hospital death occurred, which was unrelated to the surgery. The death occurred during the coronavirus disease 2019 (COVID-19) pandemic and the patient died from COVID-19 pneumonitis. Table 3 summarises the postoperative morbidity and mortality.
Discussion
Octogenarians in this study were associated with advanced aneurysm morphology and hostile neck anatomy. The CEXC device was used effectively to achieve a good seal in such hostile anatomy with satisfactory initial results.
In this study the advanced age of the patients (>80 years) was associated with an advanced disease process. The median AAA diameter was 60 mm and the infrarenal beta angulation was 80°. This hostile aneurysm anatomy in the advanced age group represents a challenge for treatment. Similarly, Lange et al 9 on behalf of the EUROSTAR collaborators found that octogenarians tend to have more advanced disease than younger patients, especially a larger diameter of the aneurysm neck and a higher incidence of angulation of the neck or iliac arteries. However, the use of the CEXC device in these patients overcomes these problems and has demonstrated a high technical success rate. In this single-centre study of octogenarians treated with this novel device, the initial technical success rate was 95%, with one type Ia EL noted on the completion angiogram which persisted on the 30-day postoperative CT scan. However, this has been successfully treated with prolonged balloon moulding of the proximal end of the graft, done as a separate procedure under local anaesthetic 103 days after the EVAR (Figure 3A–C).
High technical success rates in patients with hostile anatomy managed with the conformable Gore device were also reported by Bonvini et al,10 Lee et al11 and Mascoli et al.12 However, these reports included a small number of patients (5, 24 and 25, respectively). A study by Zuidema et al13 which included 64 patients also showed a 100% technical success rate using the CEXC device. In these four studies, no type Ia EL, type III EL or migration was seen. However, Lee et al11 reported one type Ib EL at 3 months. These results are comparable to those of the current study.
In the current study, the incidence of type II ELs on the 30-day postoperative CTA was 30%, which is similar to that observed by Lee et al (29%).11 This rate is also comparable to the incidence of type II ELs (39%) reported in a large systematic review that explored the use of different types of endoprostheses.14
We reported four postoperative complications: ischaemic colitis, unilateral buttock claudication secondary to internal iliac artery embolisation to treat a concomitant CIA aneurysm, intermittent claudication secondary to femoral artery dissection and superficial groin surgical site infection. All of these complications were managed conservatively. This 20% risk of 30-day morbidity is slightly higher than that reported in a meta-analysis by Antoniou et al15 who reported a 15% rate of 30-day morbidity in patients with hostile anatomy using different types of endografts.
The slightly higher rate of complications in our series was expected as we included patients with advanced age and multiple comorbidities. However, our results were similar to those from the EUROSTAR data, which showed that the complication rate was somewhat higher among octogenarians compared with that among younger patients.9 A similar pattern was also reported by Alberga et al16 who observed a statistically significant higher rate of major complications after EVAR in octogenarians compared with non-octogenarians.
Similar to the findings of Bonvini et al,10 no aneurysm-related secondary interventions were required in the 30-day postoperative period, which indicates that this graft may be able to achieve seal of the whole landing zone. Subsequently, it may avoid early re-interventions. However, longer term follow-up is required to determine the rate of secondary intervention in the long term. In this study one patient required re-intervention for a type Ia EL on postoperative day 103. Long-term follow-up for this graft is very scarce in the literature; one-year results for the CEXC device have been reported by Rhee et al17 who observed a 2.5% rate of re-intervention within the 1-year follow-up.
We reported one mortality, which occurred on day 28 postoperatively, and it was not aneurysm-related. This occurred during the COVID-19 pandemic, and the patient died from COVID-19 pneumonitis. Our findings suggest that the use of the CEXC device in octogenarians with severe aortic neck angulation is safe and effective within the IFU of the device.
The limitations of this study include selection bias, as patients were recruited by the consultant vascular surgeon who assessed the suitability of the device. However, all patients were discussed at MDT meetings involving multiple consultant vascular surgeons, intervention radiologists and anaesthetists. Furthermore, we did not compare the outcomes of the CEXC device with other devices or its use in a younger age group. Other limitations include the short follow-ups and the small sample size of the study, which may affect its validity. Further studies involving larger sample sizes and longer follow-ups to investigate long-term durability are warranted.
Conclusion
EVAR using the Gore Excluder CEXC system in octogenarians, particularly those with highly angulated aortic neck anatomy, is feasible within the IFU and has satisfactory initial results. However, despite these encouraging initial technical results, longer term durability needs to be investigated.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2024;3(2):65-70
Publication date:
December 15, 2023
Author Affiliations:
Department of Vascular Surgery, University Hospitals of Coventry and Warwickshire, Coventry, UK
Corresponding author:
Ashraf Elsharkawy
Consultant Vascular Surgeon,
Department of Vascular Surgery,
University Hospitals of Coventry & Warwickshire,
Clifford Bridge Road,
Coventry
CV2 2DX, UK
Email: ashraf.elsharkawy@ uhcw.nhs.uk











