ORIGINAL RESEARCH
The role of Braden scores in predicting outcomes following revascularisation for chronic limb-threatening ischaemia and their association with frailty
Baljer B,1 El-Sayed T,1 Bhullar D,1 Shelmerdine L,1 Buckley E,1 Witham M,2 Stansby G,1 Nandhra S1
Plain English Summary
Why we undertook the work: Peripheral arterial disease is a medical condition where, due to disease of the blood vessels, not enough blood reaches the leg or foot. In its most severe form, this is called chronic limb-threatening ischaemia (CLTI), which is associated with a high risk of amputation and death. Tests to measure for patients’ frailty may help predict who will do well and who will do badly after undergoing major surgery to improve blood flow to the leg, but these are not routinely used at the moment. We wanted to know if a different test such as the Braden score (a scoring system to see how at risk someone is of getting a bed sore from not moving enough in bed) could help to predict who will do better or worse after major surgery to improve the blood flow to their leg.
What we did: Patients who underwent an operation for CLTI over a 2-year period (2016–2018) in the Northern Vascular Centre, Freeman Hospital, Newcastle upon Tyne, UK were studied. The accuracy of the Braden scores and electronic Frailty Index (eFI) calculated using General Practice (GP) notes in predicting outcomes and complications of surgery were assessed.
What we found: The Braden scale was able to predict rates of amputation and complications after the operation such as infections and heart attacks. The Braden scale was also able to predict the likelihood of death following the operations. The eFI scoring system was able to predict rates of amputation, re-blockage of circulation and the need for more operations. The Braden score was found to be related to the eFI, meaning that if a patient had a worse score on the Braden scale, they were more likely to be frail on the eFI.
What this means: The Braden scale could be used as a tool to help predict outcomes of surgery for CLTI. Because the Braden scale was also found to have a link to the eFI, it could also be used as a marker for frailty on its own. However, more studies and research with larger groups of patients are needed to prove this.
Abstract
Introduction: Chronic limb-threatening ischaemia (CLTI) is a substantial healthcare burden associated with high rates of amputation, morbidity and mortality. Frailty measures have proved to be an asset in risk prediction but are not routinely universally collated. This study aimed to identify the utility of Braden scores, an assessment tool for pressure ulceration, as a prognostic indicator for patients with CLTI undergoing revascularisation.
Methods: This is a retrospective study of a prospectively maintained database of all patients with CLTI who underwent lower limb revascularisation bypass surgery between 2016 and 2018 in the Northern Vascular Centre, Freeman Hospital, Newcastle upon Tyne, UK. Their Braden scores were obtained and their electronic Frailty Index (eFI) calculated. Patients were divided into subgroups for each scoring system and their post-revascularisation outcomes were compared. This allowed us to identify the ability of these scoring systems to predict outcomes.
Results: Eighty-seven of 124 CLTI patients undergoing lower limb revascularisation bypass surgery had their Braden scores calculated. Nineteen patients (22%) had Braden scores <18 (high risk for ulceration) with a higher risk of baseline sarcopenia (32% vs 12%, p=0.04, OR 1.4, CI 0.48 to 4.53), major amputation (53% vs 28%, p=0.04, OR 1.2, CI 0.48 to 3.02), longer hospital stay (median 33 vs 14 days, p=0.04) and overall complications of pneumonia, myocardial ischaemia and wound infection (58% vs 31%, p=0.035, OR 1.8, CI 0.59 to 5.53) considered as composite factors. These patients also had worse overall survival according to Kaplan–Meier analysis (p<0.001), and the Braden scores were independently associated with death (hazard risk 1.157, CI 0.67 to 1.92; p=0.01). Braden scores were negatively correlated with an increasing eFI (p=0.016).
Conclusion: Braden scores appear to be a promising prognostic indicator of adverse outcomes in patients with CLTI undergoing revascularisation surgery. Additionally, Braden scores may also be a surrogate marker of frailty. Further larger studies are required for the validation of Braden scores and their roles in improving outcomes of CLTI intervention.
Introduction
Peripheral artery disease (PAD) poses a substantial healthcare burden worldwide as the third leading cause of atherosclerotic vascular morbidity.1 Chronic limb-threatening ischaemia (CLTI) induces rest pain and/or tissue loss and encompasses the end stage of PAD, developing in approximately 11% of PAD patients, and is associated with debilitating pain, poor quality of life, one-year limb loss (15–20%) and mortality (15–40%).2,3
Lower limb revascularisation aims to improve tissue perfusion in order to avert major adverse limb and cardiovascular events.4 The most vital index for evaluating lower limb revascularisation in CLTI is the long-term symptoms improvement and preservation of the affected limb.5 However, as evidenced by the aforementioned poor prognostic figures,2,3 this is not always possible and it is currently difficult to reliably quantify the risks and benefits of revascularisation on an individual level. Therefore, in order to aid both patients and clinicians in their clinical decision making, the development of valid risk prediction models is vital. However, concurrent models provide limited efficacy in predicting amputation and mortality.4
Several risk factors for poor postoperative outcomes have been identified, such as age and biochemical markers (including abnormalities in serum sodium, haemoglobin and low albumin).6-10 However, lone predictors have been highlighted to fail in capturing the multifactorial nature of risk in critically ischaemic vascular patients undergoing revascularisation suregry.11 Contemporary research suggests that a principal factor in universal risk determination is the frailty syndrome.11,12 Frailty has been defined as a state of vulnerability to adverse stressors due to an ageing-associated cumulative decline in physiology and homeostatic reserve.13-15 As vascular pathologies, especially CLTI, are primarily a disease of the elderly,16 the consequences of frailty are essential to consider when calculating the postoperative risks associated with lower limb revascularisation surgery. Indeed, frailty in vascular surgery patients has been shown to predict a multiplicity of poorer outcomes.17-19
Despite its recognised significance in the literature, frailty remains difficult to measure due to the absence of a gold standard index, resulting in the development of multiple assessment tools and therefore a lack of consistency.20,21 Although a variety of these models are able to identify patients at high risk of adverse events,22-24 their use in clinical practice is often limited by the need for time-consuming performance measures which are not routinely measured (gait speed, grip strength, sit-to-stand tests) or access to up-to-date primary care electronic health records (EHR).11,13,24 Furthermore, the majority of frailty measures, such as the Clinical Frailty Scale, although shown to be able to assess frailty retrospectively, are required to be measured prospectively.25
The electronic Frailty Index (eFI) for frailty assessment based on data from EHRs is the most widely used tool, particularly in primary care, and appears to be a promising predictor of adverse postoperative outcomes.24,26-28 The eFI was chosen specifically as a validated retrospective measure for our patient cohort: eFI scores are calculated using pre-diagnosed deficits that are given equal weight when categorising an individual into different frailty brackets. The eFI showed a robust predictive value for outcomes of mortality, hospitalisation and nursing home admission.24
The Braden scale is a risk assessment tool routinely collected by nursing staff for the determination of pressure ulcer risk.29 Several studies have shown that the Braden scale predicted postoperative complication rates, length of hospital stay and institutionalisation upon discharge after abdominal and pelvic surgery.11,30-34 The utility of the Braden scale in patients undergoing surgery for CLTI has not been assessed to date. This study therefore aimed to establish if preoperative Braden scores independently predict outcomes following lower limb bypass for CLTI, and whether it would be able to provide a similar predictive potential to frailty measures.
Methods
Study design and population
This retrospective analysis of a prospectively maintained database included all patients with CLTI who had rest pain and/or tissue loss for more than 2 weeks and underwent lower limb revascularisation bypass surgery only between 2016 and 2018 in the Northern Vascular Centre, Freeman Hospital, Newcastle upon Tyne. Patients’ PAD severity was classified according to the Rutherford classification. Their Braden scores were obtained prospectively by the nursing staff as described by Bergstrom et al,29 and the eFI scores were calculated retrospectively by our research team as described by Clegg et al,24 using a total of 36 variables available from primary care EHRs. The score is calculated by expressing the cumulative deficits present as an equally weighted proportion of the total.
Preoperative baseline characteristics were collected, including demographics and co-morbidities including diabetes mellitus, ischaemic heart disease, hypertension, cardiac failure, renal failure, and chronic obstructive pulmonary disease. Preoperative patients’ sarcopenia was defined through the routine preoperative CT angiogram images of each patient as a skeletal muscle area at the 3rd lumbar vertebra level of <114 cm2.35 Patients’ preoperative biochemical markers were collected including haemoglobin, leukocyte count, serum sodium, potassium, albumin, creatinine and total protein. The study was approved by the Newcastle upon Tyne Hospital Research Department and the Newcastle University Review Board.
For analysis of the Braden scale, the patients were divided into two groups: (1) scores >19 and (2) scores <18. The aforementioned values were chosen in previous studies as patients scoring >18 are deemed to be at low risk of pressure ulcers.11,31 For eFI, we followed the documented categories by the developing team: fit (0–0.12), mild (>0.12–0.24), moderate (>0.24–0.36) and severe frailty (>0.36).24
Patients were divided into subgroups for each scoring system and their post-revascularisation outcomes were compared. This allowed us to identify the ability of these scoring systems to predict outcomes. Outcomes examined were major lower limb amputation (MLLA), defined as either a below-knee, above-knee or through-knee amputation; length of stay; a composite of postoperative complications (pneumonia, myocardial ischaemia, wound infection) obtained retrospectively from clinical notes; all-cause mortality; re-intervention rates; and bypass graft occlusion. Each subgroup within the two indices was used as individual comparators.
Study outcomes
The primary outcome was the association between Braden scale and post-lower limb revascularisation outcomes (major lower limb amputation, composite of postoperative complications, all-cause mortality; re-intervention rates bypass graft occlusion and operative time as higher risk patients are likely to undergo more straightforward surgery) in CLTI patients. The secondary outcomes were the variation between different demographics and operative measures and different eFI and Braden groups, and the association between eFI and the aforementioned post-lower limb revascularisation outcomes in CLTI patients, and to study the relation between Braden score and eFI.
Statistical analysis
Patients were grouped depending on their category of frailty and Braden score. Continuous variables were tested for normality and when data were not normally distributed a non-parametric analysis and median values with interquartile range were reported. Descriptive analysis was used to calculate the mean and standard deviation of the demographic information and length of stay, and binary regression analyses were used to assess for significant correlation between the abovementioned outcomes. Kaplan–Meier survival plots were used to compare survival using a log-rank test, and multivariate analysis was performed to adjust for the key variables and risk factors. Statistical analyses were carried out using SPSS version 27 (SPSS, IBM, Chicago, Illinois, USA), and statistical significance was defined with a p value <0.05.
Results
A total of 124 patients with CLTI were included in the study (100 men (80.65%), median age 67.5 years (IQR 59–75)) with a median follow-up of 4.8 years (IQR 4.05–5.77). Their demographic characteristics, co-morbidities, biochemistry laboratory investigations, severity of peripheral arterial disease and operative time are shown as per their eFI and Braden scores in Table 1.
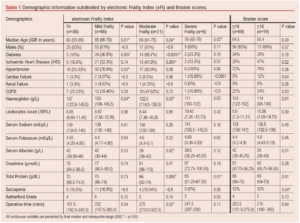
All patients underwent lower limb revascularisation bypass surgery; 66 were elective operations, 47 were urgent (within 1–5 days from admission) and 11 were emergency (<24 hours). The median operative time was 210 min (IQR 170–284). Lower limb revascularisation operations were as follows: seven aorto-bifemoral bypasses with prosthetic graft, two ileo-femoral bypasses with prosthetic graft, five femoro-femoral crossover bypasses with prosthetic graft, 16 femoral-above knee popliteal bypasses with vein graft, 67 femoral-below knee popliteal bypasses (65 with vein and 2 with prosthetic graft), 27 femoral-distal bypasses (26 with vein and one with prosthetic graft). A total of 122 patients had available information for the calculation of their respective eFIs (30 fit; 65 mild frailty; 21 moderate frailty; 6 severe frailty). Eighty-seven patients had their preoperative Braden scores documented (68 low-risk patients (score >19) and 19 high-risk patients (score <18)). The patients with a high-risk Braden score (13 men (68.42%), median age 68 years (IQR 60–76)) underwent six elective, 12 urgent and one emergency operations of one femoro-femoral crossover bypass with prosthetic graft, three femoral-above knee popliteal bypasses with vein graft, 12 femoral-above knee popliteal bypasses with vein graft and three femoral-distal bypasses with vein graft. Their median operative time was 210 min (IQR 180–300). Twelve patients (63%) required re-intervention and eight patients (42%) suffered from graft occlusion (Table 2). The principal reason for such a discrepancy in the availability of Braden scores was primarily due to the coinciding electronic uploading of inpatient notes, rendering them unavailable for the study period.
Higher risk patients with Braden scores of <18 were more highly associated with baseline sarcopenia (p=0.04, OR 1.4, CI 0.48 to 4.53) (Table 1) and stayed in hospital for a longer duration by 19 days compared with lower risk patients with Braden scores >19 (p=0.04; Table 2). Braden scores were not associated with bypass graft occlusion or re-intervention rates, but patients in the higher risk group developed a significantly higher rate of postoperative complications (p=0.04; Table 2).
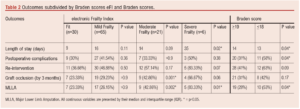
Following lower limb bypass surgery, patients with severe frailty were found to be more likely to experience longer operative time (p=0.02), while moderately frail patients had a higher graft occlusion rate (p=0.001, OR 2.8, CI 1.09 to 7.35) and the merged frail patient cohort required further re-intervention compared with the non-frail group (p=0.03, OR 1.05, CI 0.52 to 2.13). However, there was no significant association between eFI status and postoperative complications or length of hospital stay (Table 2).
Both lower Braden scores and higher eFI were associated with major lower limb amputation rates (OR 1.2, CI 0.48 to 3.02 (p=0.04) and OR 1.56, CI 0.72 to 3.36 (p=0.008), respectively). The patients’ Braden scores were negatively correlated with their eFI (correlation coefficient 0.26, p=0.016).
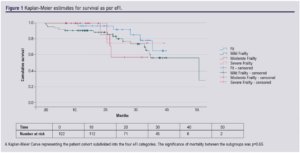
Cox regression analysis showed that lower Braden scores were associated with a higher risk of all-cause mortality, with a hazard risk of 1.157 (CI 0.67 to 1.92; p=0.01), while the eFI was unable to accurately predict mortality (Figures 1 and 2).

Discussion
Our study results highlighted that higher risk Braden scores were associated with a higher rate of MLLA, longer duration of stay in hospital, overall complication rates and all-cause mortality. Braden scores also significantly correlated with mortality.
CLTI itself is associated with high morbidity and mortality.2,3 This is more pronounced in frail patients with CLTI as they are more likely to be managed conservatively, and are associated with even higher hospital costs, mortality and amputation rates.34-38 The James Lind Alliance Vascular Research Priority Setting Partnership suggested that the foremost priority in PAD research is to identify what can be done to improve outcomes in patients with severe circulation problems to their legs.39
Our study results, in keeping with previous research,36,37 demonstrate that frailty plays a crucial role in surgical outcomes of lower limb revascularisation bypass operations. However, to our knowledge, this is the first study to examine the role of Braden scores on CLTI patients and its relation to the outcomes of revascularisation.
Our patients with Braden scale of <18 had a two-fold increased risk of limb loss and a seven-fold increase in overall mortality. This patient group demonstrated worse sarcopenia compared with their peers, which put them at a higher risk of postoperative outcomes, particularly as sarcopenia has been previously reported as an independent factor for MLLA rates and overall mortality following lower limb bypass operations.35 However, higher risk Braden scores were not statistically associated with more frequent bypass graft occlusion or re-intervention.
On the other hand, the greater rates of limb loss present among the more frail subgroups is underpinned by the presence of similarly increasing rates of bypass graft occlusion and re-intervention rates. This phenomenon is likely highly conducive of graft failure, leading to the more frequent need for MLLA. However, the underlying principle behind the failure of the grafts is not completely evident as it is likely a multifactorial cause. Nevertheless, patients with poorer eFI scoring were likely to suffer from anaemia, diabetes, ischaemic heart disease and lower total protein levels which have been linked to increased morbidity and poorer outcomes following vascular surgery.40-43 The operative surgical time was longer in moderately frail patients, which puts them at a higher risk of postoperative complications.44 Severely frail patients underwent the second shortest operations, so one might assume that they underwent more straightforward arterial reconstruction that suited their frailty.
It is important to acknowledge, however, that frailty and co-morbidity are interrelated but distinct entities, meaning that frailty is independent of disease.45,46 The Braden scale, in contrast, provides a more accurate snapshot of each individual patient at a given moment in time, and may provide further invaluable information in relation to the non-descript factors of frailty.
This notion is further emphasised by the finding that patients in the higher risk subdivision of the Braden groups were more likely to suffer from postoperative complications (Table 2). In addition, their average length of stay as an inpatient was over twice as long, which puts them at a higher risk of hospital-acquired infections.47 The finding that the eFI score was not associated with complication rates, length of stay and mortality in our patient cohort while the Braden score was efficacious in doing so, suggests that the multifactorial and temporal nature of frailty may not be fully captured by eFI, and it may be prudent to use the Braden scale as a supplementary marker for frailty in this patient population.
eFI and Braden scores were weakly negatively correlated (correlation coefficient 0.26), which could suggest that the two scales might be interconnected. However, further research is required to evaluate whether the Braden scale may be used not only as an adjunct but also as a surrogate measure of frailty, which would prove to be vital for future research due to its ease of measurement.
Several individual factors play a vital role in eventual poor surgical outcome, and therefore a risk prediction model consisting of a combination of tools would be an invaluable asset to guide decision-making. This study presents frailty as a key contributing factor to poorer surgical outcomes following primary lower limb revascularisation surgery for the treatment of CLTI. Furthermore, it highlights the potential clinical use of the Braden scale as a prediction tool for risk assessment in lower limb revascularisation, especially with regard to MLLA rates. This suggests that frailty and Braden scores may both hold a place in the aforementioned risk prediction model. However, as the current results also suggest that Braden scores may be interchangeable with frailty scores, only one of these tools would be needed in the model. Due to its accessibility, Braden scores may be best placed in this model. The use of such models should not prevent patients from receiving lower limb bypass operations, but aid in the risk evaluation process on an individual patient level. This will allow for joint decision making with regard to whether primary revascularisation or primary MLLA is appropriate, to ultimately improve outcomes.
Study limitations
The limitations of this study include the relatively small sample size from a single centre with a relatively homogenous study population, all of whom are based in the North-East of England.
Conclusion
This UK Vascular Surgery tertiary centre study shows that the Braden scale is associated with MLLA and complication rates, overall mortality and length of hospital stay following lower limb revascularisation in patients with CLTI. Braden scores have the potential to be used as risk stratification tools in the context of CLTI. Furthermore, as the Braden scale has been independently linked to surgical outcomes and correlated with the eFI, it may be a surrogate marker for frailty. Further studies in a patient cohort are recommended for the validation of Braden scores and their role in improving outcomes of CLTI intervention.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2023;2(2):76-82
Publication date:
February 7, 2023
Author Affiliations:
1. The Northern Vascular Centre, Freeman Hospital, Newcastle upon Tyne, UK
2. NIHR Newcastle Biomedical Research Centre, Newcastle University, UK
Corresponding author:
Tamer El-Sayed
Clinical Lecturer in Vascular Surgery, The Northern Vascular Centre, Fourth Floor, Freeman Hospital, Freeman Road, High Heaton, Newcastle upon Tyne, NE7 7DN, UK
Email: [email protected]











