ORIGINAL RESEARCH
Trends in lower limb deep vein thrombosis and associated deep venous procedures across the United Kingdom from 1998 to 2022
Machin M,1 Khatri A,1 Salim S,1 Gwozdz AM,1 Lim C,2 Davies AH,1 Shalhoub J1
Plain English Summary
Why we undertook the work: In the UK there are ongoing efforts to reduce the number of blood clots (deep vein thrombosis, DVT). Despite these initiatives, around 35,000 people in the UK suffer from DVT each year. In the short term, DVT can go on to cause blood clots in the lungs, known as a pulmonary embolism; this can impair the body’s ability to pass oxygen to the blood stream. In the longer term, DVT can cause post-thrombotic syndrome (PTS), which is a term used for the collection of symptoms such as swelling, pain and other long-term issues that occur in up to 50% of those affected by DVT. A potential way of shortening the recovery time and reducing the chance of developing PTS is to have the blood clot removed through a procedure known as early thrombus removal. This can be achieved by open surgery or minimally invasive keyhole procedures such as clot-busting medicines (thrombolysis), catheter-based suction/aspiration techniques, or catheter-based mechanical removal techniques. For established PTS, the scarred and narrowed veins can be treated with stretching of the vein (balloon venoplasty) and placement of a stent. These are referred to as interventions for chronic disease. Since the introduction of these techniques there has been conflicting evidence from clinical studies and a lack of certainty in the scientific community. For the UK, there are no up-to-date reports on the number of people suffering from DVT or PTS, or the number of procedures performed to treat these conditions. These findings are important as they will provide us with a barometer of current practice across the UK and may provide a strong rationale for further clinical studies.
What we did: To identify how many cases of DVT and PTS were diagnosed and how many procedures were performed, we searched the national database published by NHS England. From this, we extracted information for the period from 1998 to 2022. We undertook statistical tests to assess if the trends in the data were significant. From this, we gain an appreciation for the clinical care being provided and the potential impact of this on the healthcare service.
What we found: The rate of hospital admission for DVT has decreased since 1998. This may be because hospitals are now increasingly likely to see patients in same-day assessment units, such as ‘ambulatory care’, rather than admitting them to hospital. There was a single year in which the cases of DVT were much higher, coinciding with the COVID-19 pandemic. The rate of procedures undertaken for early thrombus removal after DVT is increasing year on year. The rate of procedures for chronic venous disease had been increasing up until the year 2014, after which they have levelled off.
What this means: The increase in procedures for early thrombus removal and for established chronic venous disease may indicate that specialist venous services are likely to be required to deliver this care consistently. This cannot be explained by a rise in cases of DVT or PTS alone. However, the recent plateau in the rates of interventions for established chronic venous disease suggests that this practice is still in its early phases of adoption and its widespread use may be limited by the lack of clear evidence in this field.
Abstract
Introduction: Following acute iliofemoral deep vein thrombosis (DVT), early thrombus removal may reduce the risk of subsequent post-thrombotic syndrome (PTS). For those with established chronic venous disease (CVD) secondary to obstructing iliac vein lesions, venoplasty and venous stenting can be used to restore patency. Currently, these interventions are associated with significant costs with a lack of clear evidence to support their use, hence an understanding on the trends of intervening is required. This study assesses overall trends in diagnoses of DVT and PTS resulting in hospital admission, alongside the number of deep venous procedures performed across the UK from 1998 to 2022.
Methods: National database analysis was undertaken using Hospital Episode Statistics (HES) data from NHS England from 1998 to 2022. The number of primary diagnoses for DVT and PTS episodes resulting in hospital admission, in addition to primary procedure codes for open and percutaneous deep venous procedures, were extracted for analysis. Rates of DVT and PTS episodes were analysed to contextualise the rates of deep venous procedures undertaken. The Mann–Kendall test was undertaken to assess for trends within the data.
Results: There has been an overall significant (p<0.05) downwards trend in admissions for DVT between 1998 (33,205) and 2022 (29,831). There was an isolated peak in cases coinciding with the coronavirus pandemic, at which time numbers increased 2.4-fold. Diagnoses of PTS as the primary diagnosis for admission have significantly risen (p<0.05) but overall reported numbers were low across all time points (range 19–446). Regarding acute deep venous interventions, the number of percutaneous venous thrombus removal procedures demonstrated a significant upward trend (p<0.05), with peak cases in 2019 of 451. Percutaneous transluminal venous thrombolysis was the most common procedure performed across all time points. Percutaneous mechanical or aspiration venous thrombectomy procedures also significantly increased (range 7–113, p<0.05), and demonstrated the most persistent upward trend for a given technology. Regarding interventions for chronic disease, percutaneous venous stenting and venoplasty procedures have shown a significant upward trend since 1998 (range 0–1,469, p<0.05). Numbers increased to a peak of 1,469 in 2009, after which the gradient has levelled and potentially plateaued.
Lessons learnt: Considering these increasing practices, contemporary randomised controlled trials are required to provide certainty of effectiveness.
Conclusion: The number of early thrombus removal procedures, mainly catheter-directed thrombolysis, continues to increase despite the backdrop of conflicting evidence supporting its use. This does not coincide with an increase in hospital admissions for DVT. Interventions for iliac-obstructing CVD, including venoplasty and stenting, dramatically increased but have since plateaued.
Introduction
Deep vein thrombosis (DVT) affects as many as 35,000 people per annum in the UK.1 DVT causes substantial morbidity, with as many as 50% of those affected developing post-thrombotic syndrome (PTS), characterised by lifelong leg pain, oedema, skin changes and ultimately venous ulceration.2,3 PTS results in reduced quality of life (QoL) and an overall disability burden comparable to chronic obstructive pulmonary disease or heart failure.4
PTS is thought to develop due to venous obstruction and/or valvular reflux following the pathophysiological process of DVT resolution, with conversion of fibrin to collagen (ie, scarring),5 subsequently leading to venous hypertension.6 Those with iliofemoral DVT are more likely to develop PTS than those with distal DVT, hence they represent a key cohort of patients when considering treatments in the prevention and subsequent treatment of PTS.7
Deep venous practice has changed dramatically over the past three decades with the introduction of catheter-directed thrombolysis (CDT) for early thrombus removal and the use of venous stenting for restoring patency in chronic iliac disease (circa 1994).8,9 Percutaneous catheter-delivered pharmacomechanical thrombectomy and non-lytic mechanical thrombectomy are now clinical realities10-12 alongside the widespread availability of dedicated venous stents.13-15 These interventions are offered within the National Health Service (NHS) in Trusts offering complex venous services with designated acute and chronic clinical pathways.16
Despite this, there is still a lack of clarity regarding the evidence to support early thrombus removal in acute iliofemoral DVT alongside a complete lack of comparative randomised evidence to support venous stenting for chronic venous outflow obstruction.17,18
Illustrating this lack of clarity with respect to acute thrombus removal is the National Institute for Health and Care Excellence (NICE) guidelines stating that CDT for symptomatic iliofemoral DVT can be ‘considered’.19 The European Society for Vascular Surgery 2022 Clinical Practice Guidelines reiterate this lack of clarity, suggesting ‘consideration’ for selected patients with a class 2a recommendation.20 The result of this uncertainty is that deep venous practice is in a state of flux.
Lim et al previously reported an increasing trend in percutaneous deep venous procedures between 2005 and 2015, with open deep venous procedures declining and remaining persistently low.21 Contemporarily, the PTS after Catheter-directed thrombolysis for deep Vein Thrombosis (CaVenT) study, published in 2012, had reported very promising results supporting the use of early thrombus removal in preventing PTS.22
However, seven years, two further pivotal randomised controlled trials (RCTs)23,24 and the global COVID-19 pandemic have since passed. The Acute venous Thrombosis: Thrombus Removal with Adjunctive Catheter-directed Thrombolysis (ATTRACT) trial, published in 2017, cast doubt on the benefits of early thrombus removal in the prevention of PTS.24 Additionally, the ultrasound-accelerated CAtheter-directed thrombolysis Versus Anticoagulation for the prevention of post-thrombotic syndrome (CAVA) trial, published in 2020, failed to demonstrate any benefit in reduction of PTS or quality of life.23
Furthermore, although commercially sponsored registries report promising results, there is still yet to be any comparative evidence on the use of venous stenting in chronic occlusive ilio-caval venous disease.13,14
The nuanced and complex interpretation of RCTs investigating early thrombus removal in acute DVT, alongside the sparsity of evidence for chronic deep venous interventions, means that there has been a lack of clarity over the last decade.
Aims and hypothesis
Using the Hospital Episode Statistics (HES) from 1998 onwards reported by NHS England, we aimed to provide a contemporary report on the trends in the number of primary open and percutaneous procedures for deep venous pathology and the rates of hospital admissions for DVT and PTS (which are the main clinical indications for these procedures) in England from 1998 to 2022. We hypothesised that the rates of deep venous procedures across the UK are decreasing. This is a critical period spanning publication of pertinent RCTs and key registries investigating the use of deep venous procedures in the prevention and treatment of PTS.
Methods
The HES database, containing information relating to all inpatient hospital admissions, outpatient appointments and emergency department attendances within the NHS, was searched for hospital admissions with primary diagnoses of DVT and PTS as well as for primary deep venous procedures on 29 September 2022. Records were searched from 1998 up until 2022, spanning a 24-year period.21
The data processing pathway has previously been described and is available through NHS Digital.21,25 The HES database is published per financial year, generated from regular data submissions by healthcare providers to NHS commissioners for financial reimbursement. NHS Digital processes the data to allow for secondary analysis for national monitoring and research purposes.
Each hospital episode, defined as a finished admission under the care of a named consultant within an NHS Trust, is represented once within the database, with the primary diagnosis requiring admission and the most resource-intensive procedure being selected. Ad hoc data quality checks are performed, and investigations based on information supplied by providers or from feedback from HES data users.25
Diagnoses and procedures are recorded according to the International Classification of Disease 10th Edition (ICD-10) and Office of Population Censuses and Surveys Classification of Interventions and Procedures version 4 (OPCS-4) codes, respectively. No unique diagnostic code for non-thrombotic iliac vein lesions (NIVLs) exists. Regarding nomenclature, when referring to interventions for iliac-obstructing CVD, this includes chronic PTS and NIVLs.
Data handling and analysis
Appendix 1, supplementary Table 1 (online at www.jvsgbi.com) shows the ICD-10 codes used for the extraction of data relating to hospital admissions with a primary diagnosis of DVT. In addition, the data pertaining to admissions with a primary diagnosis of PTS (ICD I87.0) were also extracted. Appendix 1, supplementary Table 2 (online at www.jvsgbi.com) shows the OCPS-4 codes used to identify hospital admissions with relevant primary deep venous procedures, as well as the groupings of similar primary procedures for analysis.
Analysis included simple descriptive statistics in addition to Mann–Kendall trend testing. The Mann–Kendall test was undertaken in R using the Kendall library. A p value of <0.05 was set as the level of significance, with additional Holm–Bonferroni correction (Holm’s sequential Bonferroni procedure with ranking of p values) for multiple testing. Parametric assumptions could not be fulfilled, hence the Mann–Kendall test was deployed in place of linear regression. Locally Weighted Scatterplot Smoothing (LOWESS) was also undertaken.
Results
Rates of hospital episodes with primary diagnosis of DVT
Figure 1 illustrates the number of hospital admissions within the NHS with a primary diagnosis of DVT between 1998 and 2022. There was an overall reduction in total admissions for DVT between 1998 (33,205) and 2022 (29,831), with a significant downwards trend (tau=–0.39, p<0.05); LOWESS illustrates this (Appendix 2, supplementary Figure 1 – online at www.jvsgbi.com).
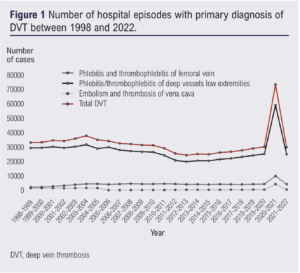
Across the year 2020–2021 there was a sharp increase in episodes of DVT (n=73,413), 2.4-fold greater than the baseline rate from 1998–2020 (range 24,411– 37,926).
Embolism and thrombosis of the vena cava as an indication for admission has varied substantially but overall remained static since 1998 (range 64–4,299, p=0.50). There was an abrupt decline in the rate of inferior vena cava (IVC) thrombosis from the year 2004–2005, since when there has been a steady increase from 2005 onwards up to 2020. Similar to DVT of the lower extremity veins, thrombosis of the IVC increased dramatically in the year 2020–2021, 5.8-fold greater than the baseline rate from 1998–2020 (range 164–1,511) (Appendix 2, supplementary Figure 2 – online at www.jvsgbi.com).
Rates of hospital episodes with primary diagnosis of post-thrombotic syndrome (PTS)
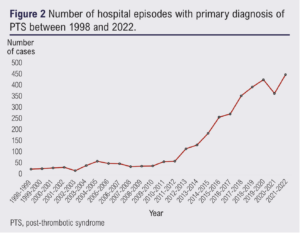
Figure 2 shows that admissions for PTS have significantly risen from 1998, particularly after 2012 (tau=0.84, p<0.05). The overall reported numbers of hospital admissions for PTS were low across all time points (range 19–446).
Rates of hospital episodes with primary procedure of early thrombus removal procedures
Numbers of percutaneous venous thrombus removal procedures demonstrate a significant upward trend from 1998–2022 (tau=0.85, p<0.05; Figure 3). There were no cases of percutaneous early thrombus removal until 2006. Since 2019, total numbers of percutaneous early thrombus procedures have levelled off and slightly reduced (not significant), from a peak of 451 across the year 2019–2020 to 323 episodes per year contemporarily.
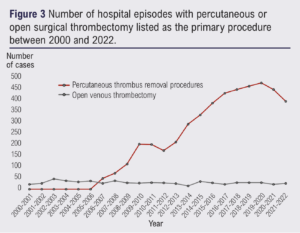
Percutaneous transluminal venous CDT was the most common procedure performed across all time points for early thrombus removal, the trend of which mirrored the overall increasing trend for percutaneous early thrombus removal (range 0–349, tau=0.68, p<0.05). Percutaneous mechanical or aspiration venous thrombectomy procedures have also significantly increased (range 7–113, tau=0.80, p<0.05), and demonstrate the most persistent upward trend for a given device technology, although it does remain less frequent than CDT interventions.
Open venous thrombectomy numbers continue to remain low and static (range 14–44, p>0.05). Percutaneous thrombolysis with reconstruction (ie, early thrombus removal with adjunctive stenting) demonstrated a significant upward trend (0–99, tau=0.67, p<0.05), mirroring that of early thrombus removal.
Rates of hospital episodes with primary procedure of percutaneous deep venous interventions without mention of thrombectomy/thrombolysis
Figure 4 demonstrates the number of hospital episodes with primary procedures pertaining to percutaneous endovenous reconstruction with venoplasty and venous stenting (interventions used for chronic disease).
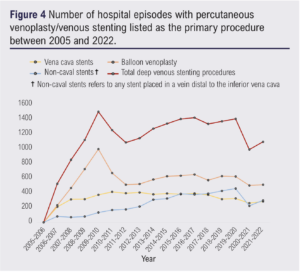
Total percutaneous venoplasty and venous stenting procedures (without mention of thrombus removal or thrombolysis) demonstrated a significant upward trend since 1998 (range 0–1,469, tau=0.38, p<0.05). There were no cases of percutaneous endovenous reconstruction prior to 2006. Numbers increased up to a peak of 1,469 in 2009, after which the gradient has levelled and potentially plateaued. Percutaneous venoplasty (without mention of stenting) peaked at 972 cases in 2009 and, since 2014, numbers have remained relatively static (range 0–972). Since 2005 the trend has not significantly changed.
Stenting procedures without mention of thrombolysis (ie, for chronic disease) demonstrated a significant increasing trend from 2005 (range 0–776, tau=0.63, p<0.05).
Discussion
We present the most comprehensive report on the current status of deep venous interventions in the UK using data from a national registry over a 24-year period.
We have demonstrated a significant downward trend for the rate of hospital admissions for DVT since 1998. This is unsurprising given the change in patient pathways for the investigation and management of DVT, with hospitals offering ambulatory pathways avoiding the need for admission. The rate of DVT dramatically increased across the year 2020–2021, which corresponded to the peak incidence of infections during the COVID-19 pandemic in the UK.26 The incidence of DVT in those testing positive for COVID-19 has been demonstrated to be as high as 16%, coinciding with the 2.4-fold increase reported in our analysis.27 Reassuringly, the rate of DVT returned to within the baseline range the following year.
Rates of IVC thrombosis remained unchanged within the study period. However, there was a step-like reduction in 2005, from which there has been a slow increase each year (non-significant). We explored the impact of the number of admissions for IVC filter placement over this time period; however, the decline appears unrelated as the number of IVC filter placements had not significantly increased between the years 2004 and 2006. It is currently unclear as to what caused this change in coded IVC thrombosis episodes; this cannot be explained by changes in coding descriptors (ie, ICD-9 to ICD-10).
The rate of admission for PTS reported in this study is incredibly low, likely reflecting the underutilisation of diagnostic coding for this condition, with use of chronic venous insufficiency or venous ulceration instead. Epidemiological data on PTS are generally poor given the difficulties in reaching a standardised diagnostic criterion. The increase reported in this study is likely to reflect increased utilisation of the PTS code rather than a true increase in incidence, but we lack evidence to support this.
This study demonstrated a significant increasing trend of early thrombus removal procedures performed in the NHS which cannot be explained by an increase in hospital admissions for DVT. The bulk of evidence for early thrombus removal consists of three large RCTs investigating whether early thrombus removal in acute proximal DVT using CDT reduces risk of subsequent PTS.22–24,28,29 The CaVenT study, published in 2012, was the only study of the three key RCTs to report a significant difference in favour of CDT for PTS prevention.22 This significant finding corresponds with an increasing rate of percutaneous venous thrombus removal procedures within the NHS from 2011 to 2017.
Subsequent publication of the ATTRACT trial in 201724 and the CAVA trial in 202023 then cast doubt on its efficacy. Both of the trials produced a negative result. The gradient of the increase for percutaneous venous thrombus removal procedures levelled off from 2017 onwards, corresponding to this conflicting evidence. Regression analysis of the results up to the COVID-19 pandemic demonstrated a similar finding, suggesting that the stimulus for the plateau occurred prior to the pandemic.
However, subgroup analysis of those participants in the CAVA trial who achieved successful recanalisation showed a significant reduction in the severity of PTS, suggesting appropriate patient selection and improved endovascular protocols may show a potential benefit of CDT.28 The above trials have suffered heavy criticisms for methodological weaknesses, detailed elsewhere in the literature.30,31 Despite an upward trend, the lack of clear evidence to support its use is likely stemming the floodgates from widespread adoption of early thrombus removal within the NHS.
An interesting observation is that open surgical thrombectomy is rarely performed, but its trend has not significantly altered. Open surgical thrombectomy tends to be performed as an emergency for phlegmasia cerulea dolens rather than for the risk reduction of subsequent PTS. Previously, percutaneous procedures would often not be able to restore patency immediately, explaining why percutaneous measures have not completely replaced open thrombectomy but supplemented it for a different clinical indication. However, the effect of contemporary thrombectomy devices such as AspirexTM (BD, New York) and ClotTrieverTM (Inari, Caifornia) catheters have yet to be seen.
The rate of interventions for iliac-obstructing CVD seems to have plateaued. A potential explanation could have been the COVID-19 pandemic; however, regression analysis at earlier time points demonstrated similar results. This practice is still within a phase of early adoption, hence the overall number remains low. There remains a lack of high quality RCT data relating to the use of percutaneous deep venous stenting or venoplasty for established CVD, reflected in the lack of recommendations by current NICE guidelines. A previous systematic review reported non-randomised evidence which supported the use of venous stenting;18 however, the authors noted the quality of the literature to be poor, limiting its findings.
A single RCT exists in the literature, containing as few as 58 participants. The trial randomised patients to either iliac vein stenting or best non-stenting (medical) therapy.32 A significant improvement in severity of disease, as assessed by the Venous Clinical Severity Score, was reported. The trial had several limitations including being a single-centre trial with small numbers, of which only 16 of the 51 participants had PTS. Publication of a large externally valid RCT will likely impact the rates of percutaneous deep venous stenting in the future. The C-TRACT (USA) and BEST (UK and Europe) trials are currently underway, which should provide grade A evidence to guide this practice in the future.33
Limitations
There are several limitations in the interpretation of this analysis. HES data are collated from service providers as part of the Commissioning Data Set and are used for monitoring and reimbursement. These data are not collected primarily for research purposes and their accuracy cannot be assured.
Data included in this study relate only to admitted patient care. Because of this, we are unable to comment on overall trends of DVT and PTS episodes which would include those treated via ambulatory or outpatient pathways. Hence, all conclusions drawn are in relation to admitted patient care.
Furthermore, combined procedures are often performed within one hospital episode, but only one – that which is most resource intensive – is represented as a single primary procedure code. For example, a procedure following acute DVT may include thrombolysis, venoplasty and deep venous stenting; however, it is likely to be coded with thrombolysis as the primary procedural code, meaning that the whole picture is not represented. Therefore, using primary procedures within HES data is likely to under-report the number of procedures undertaken. Similarly, the number of admissions with a diagnosis is also likely to be under-represented in a similar manner, but also due to the HES database not capturing episodes of DVT and PTS managed without hospital admission.
Although a primary procedure performed is often associated with a specific presentation, it is not possible to ascertain this as the indication for a procedure is not recorded within the HES data.
Lessons learnt
The rate of patients admitted for DVT has been slowly down-trending since 1998. This is likely due to change in patient pathways and a move to ambulatory management of DVT.
Despite a decline in admissions for DVT, there has been an increase in early thrombus removal procedures. Considering the increasing acute and chronic interventional practices across the UK, additional dedicated complex venous services might be required in the future to deliver this care consistently. Achievable and affordable complex venous services with defined acute DVT and CVD clinical pathways have been demonstrated in the NHS.16 The lack of certainty in the evidence, alongside lack of clinical expertise, is likely preventing national widespread adoption of this model.
Conclusion
Although rates remain high, hospital admissions for DVT have been significantly declining since 1998. The number of early thrombus removal procedures, mainly CDT, continue to increase despite the backdrop of conflicting evidence supporting its use. Interventions for iliac-obstructing CVD, including venoplasty and stenting, dramatically increased but have since plateaued, suggesting that this practice is still in the early phases of adoption and its widespread use may be limited by a current lack of comparative evidence.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2023;2(3):160-166
Publication date:
May 5, 2023
Author Affiliations:
1. Department of Surgery and Cancer, Imperial College London, London, UK
2. Department of Vascular Surgery, Royal Free London NHS Foundation Trust, London, UK
Corresponding author:
Matthew Machin
Academic Clinical Fellow in Vascular Surgery, Imperial College London, London, W6 8RF, UK
Email: matthew.machin12@ imperial.ac.uk











