ORIGINAL RESEARCH
Tissue sampling technique for diabetic toe amputations: a survey of current UK practice
Devangi M,1 Juszczak MT,2,3,4 Sam R,2,3 Tiwari A,2,3,5 Murray A,2,3 Wall ML1,3,4
Plain English Summary
Why we undertook the work: Infection of the bones and soft tissue of the foot in patients with diabetes is a growing problem across the UK, which can be difficult to treat. It is important to obtain accurate samples to identify the bacteria causing the infection to help guide treatment. We wanted to assess the techniques consultant surgeons deployed to gain accurate samples and what training they had received for this important step in the patient’s care. This is important because guidelines are available but are not consistent.
What we did: An online survey was designed to assess consultants’ knowledge, experience and training in the management of patients with diabetic foot infection and to evaluate their practice. We were particularly interested in what they actually did in theatre, what samples they sent to the laboratory and how the results of these samples affected the future management of patients. The survey was emailed to individual surgeons who were members of the national UK Society of Vascular Surgeons.
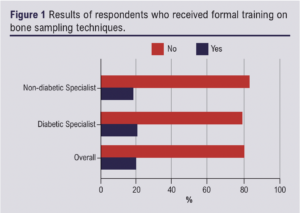
What we found: Responses were received from 109 surgeons from 13 regions across the UK. This is a response rate of 24%. Only one in five respondents had received formal training in taking samples for diabetic foot disease and only 60% of respondents said they always or usually took samples for infected diabetic foot disease. Finally, only 29% of respondents said their unit had a formal policy guiding sampling.
What this means: There is huge variability across the UK in taking samples to identify bacteria in diabetic patients presenting with foot infection. Training and formal policies to guide surgeons are seriously lacking. These deficiencies probably mean patients are not receiving antibiotics targeted specifically at the causative bacteria and need addressing urgently.
Abstract
Background: The management of diabetic foot disease remains a challenge for clinicians. Outcomes are improving with the development of integrated diabetic foot teams. Understanding tissue sampling/processing and a standardised evidence-based approach would help develop integrated pathways to improve patient care and outcomes.research priorities. This paper presents the results of this process.
Aims: In collaboration with the Vascular Research and Innovation Consortium (VaRICS), this study aimed to evaluate the knowledge base of UK vascular consultant surgeons regarding sampling and processing techniques for diabetic foot disease.
Methodology: An online questionnaire was designed to assess consultants’ knowledge levels of surgical techniques, infection control, placement of interventions, sampling techniques and processing. This was then distributed to vascular surgery consultants via the Vascular Society of Great Britain and Ireland (VSGBI) email newsletter.
Results: 109 consultants participated in this study, of which 63 (58.9%) had a sub-specialist interest in diabetic foot disease. Only 21 (19.6%) consultants had received formal training in bone sampling techniques. Eighteen (62.1%) of the respondents reported having received training at the departmental level. An average of 10–20 toe amputations were performed per month in most units. In 37% of cases, samples from clean bone were always taken after wound cleaning and debridement following toe amputations. A majority of samples from toe amputations consisted of proximal bone (64.5%), pus swabs (51.4%) and tissue/toe resections (45.8%). Most of the contamination reduction measures included saline washing and changing instruments between samples. The majority of consultants (85.6%) are unaware of the microbiology processing techniques. Conclusion: This study shows that, across the nation, in the management of diabetic patients presenting with foot infection, there is huge variation in training, sampling practice/policies and management pathways. These problems need to be urgently addressed.
Introduction
The increase in the prevalence of diabetes mellitus and its associated complications has become a major public health issue.1,2,3 Foot-related complications affect 2–2.5% of people with diabetes, equating to a point prevalence of approximately 58,000 people in England alone.3
The management of diabetic foot disease is complex, involving input from a foot protection team and a multidisciplinary team (MDT) of professionals.4 Surgical debridement of infected tissue and antimicrobial therapy to manage infection are the two mainstays of therapy for acute foot infections. The surgical aspect of the management of diabetic foot disease and the importance of appropriate training for vascular surgeons has been acknowledged by the Vascular Specialty Advisory Committee and the General Medical Council. The management of fulminant diabetic foot sepsis is one of three critical conditions in the new vascular surgery syllabus, revised in August 2021.5 Alongside surgical debridement, antimicrobial therapy for managing residual infection should be guided by microbiology sampling and cultures, although very few papers report on sampling and processing techniques. Culture results are dependent on sampling method, and the International Working Group on the Diabetic Foot (IWGDF) recommends obtaining samples for culture aseptically. This should be done before or close to the start of antimicrobial treatment, to enable empirical treatment to be amended appropriately.4,6,7
This study aimed to determine the training, practice and level of knowledge that UK vascular surgical consultants have regarding techniques for tissue sampling and processing in diabetic foot infection and to identify the prevalence of unit policies for managing diabetic foot infection. This is important because there are variations in the current guidelines available. For example, IWGDF recommends 6 weeks of antibiotics in patients who do not undergo complete resection of infected bone whereas, in patients in whom all infected bone has been resected, antibiotic treatment should not be needed for more than 1 week.6 On the other hand, the National Institute for Health and Care Excellence (NICE) recommends offering prolonged antibiotic treatment (usually 6 weeks) according to local protocols.4
Methodology
The steering group for this study consisted of three consultant vascular surgeons, a research fellow in methodology, a specialty trainee and a foundation doctor. The steering group coordinated the questionnaire development, validation, distribution and analysis.
Questionnaire development
Survey questions for each of the three domains (demographics, sampling, results interpretation) were discussed by the steering group with a focus group of vascular consultants across two tertiary referral vascular centres. All decisions required unanimous agreement amongst the focus group members. Additionally, questions were reviewed to ensure wording was succinct, clear and unambiguous by our research methodologist. The CHERRIES checklist was used to assist the designing of the questionnaire.8
Questionnaire validation
A preliminary survey of 31 questions across the three domains, including a range of binary, multiple choice and free-text open ended questions, was designed. This was validated through two rounds of piloting across two tertiary vascular centres with 10 consultants. After round 1, major (questions removed or added) and minor alterations (wording alterations) were made based on the feedback from the consultant body. Alterations were made in alignment with recommendations from our research fellow.
Questionnaire distribution
The online survey was disseminated using https://www.onlinesurveys.ac.ukTM software. Demographic data collection included background information (years of consultant practice, number of procedures performed per unit, population served by unit) and the presence of a diabetic foot sampling policy within the unit (NVR submission, sampling technique policy). Participants were advised that their responses would be anonymised.
The final survey (Appendix 1 online at www.jvsgbi.com) was distributed from January 2022 to April 2022 via email, inviting approximately 370 consultant members of the Vascular Society of Great Britain and Ireland (VSGBI) to participate. The study was publicised on the VSGBI, the Rouleaux Club and Vascular and Endovascular Research Network (VERN) twitter feeds to encourage all consultants to complete. Three months were allowed for responses with a reminder email circulated at 4 and 8 weeks.
Data analysis
Data were analysed by the steering group using Microsoft Excel. Where data were missing, they were not analysed; however, the majority of questions were completed. Optional questions such as additional comments were not answered frequently within the study.
Results
Demographics
The survey received 109 responses, of which 63 (58.9%) declared that they had a specialist interest in diabetic foot disease. Responses were received from 13 deanery regions (Appendix 2, Graph 1, online at www.jvsgbi.com). The duration of consulting experience and the unit size covered by the survey are included in Appendix 2 (Tables 1 and 2, online at www.jvsgbi.com), with 58 (54.2%) stating that their units perform 10–20 minor amputations per month (Appendix 2, Table 3, online at www.jvsgbi.com). A total of 23 (21.3%) respondents stated that their unit enters minor amputations on the National Vascular Registry, and 31 (29%) respondents stated that their unit does have a formal policy on diabetic foot sampling.
Training
Only 21 (19.6%) respondents had received formal training in diabetic foot treatment (Figure 1). Of these, 18 (62.1%) reported having received training at a departmental level (see Appendix 3, Tables 1 and 2, online at www.jvsgbi.com).
Sampling and processing
Figure 2 shows the specimens routinely sent in theatre during toe amputations. Sixty-nine (64.5%) respondents declared they always took a proximal bone specimen. Of the respondents who sent specimens, 51 (60%) sent more than one sample at the time of sampling from each incision, with 40 (37%) stating they always clean the operative field after amputation but before sending specimens.
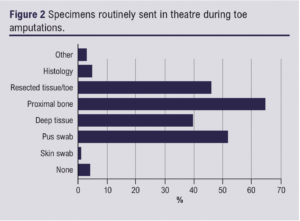
The steering group gave options on nine different techniques to reduce contamination, with the preferred technique being saline wash (58, 63.7%). The preferred technique for bone sampling reported was use of Rongeurs (71, 78.9%) with 88 (88%) reporting that they sent their samples dry. Only one respondent contacted the laboratory to inform them of the imminent arrival of a specimen, whilst 41 (39.4%) did not know how long specimens took to get to the laboratory. Fifty-one (49.1%) respondents who were informed felt their microbiological specimens were at the laboratory within 4 hours of sampling. Only five (4.7%) respondents sent specimens for histology. The majority of respondents (89, 85.6%) were unaware of the processing technique of their samples upon arrival at the laboratories and 58 (55.8%) respondents reported an expectation of a result within 49–72 hours. A further breakdown of sampling techniques is shown in Appendix 4 online at www.jvsgbi.com.
The majority of respondents stated that positive cultures change their management of the patient (Appendix 5, Table 1, online at www.jvsgbi.com). Respondents had varying opinions on the impact of positive proximal bone cultures on patient management when compared with other sample types. Specifically, 88 (93.6%) respondents stated that a positive proximal bone culture would change patient management compared with 35 (36.1%) for pus swab, 62 (68.1%) for deep tissue, 36 (39.1%) for resected non-proximal bone and 16 (20.3%) for histology. In the presence of proximal bone infection, 76.5% of respondents would treat with antibiotics for 6 weeks as opposed to 6 (6.3%) if a pus swab came back positive (Appendix 5, Table 2, online at www.jvsgbi.com). The differing sampling techniques, the rate of wound closure, length of hospital stay and follow-up routines are shown in Appendix 5, Tables 3–10, online at www.jvsgbi.com.
Discussion
The importance of diabetic foot disease treatment development was recently highlighted in the VSGBI James Lind Alliance Priority Setting document.9 It emphasised the importance of improving treatment to improve wound healing rates and prevent further amputation. Diabetic foot disease presents the clinician with significant therapeutic challenges, with both medical and surgical approaches that can be undertaken in its management. At the point of surgical intervention, the clinician is provided with an opportunity to gather a large number of samples from various different tissues of the diabetic foot. A standard level of training is not specified in the intercollegiate surgical curriculum, and while guidance is available, it is inconsistent. This study provides evidence of the variation in sampling practice among vascular surgeons within the UK and therefore the difficulty in establishing guidelines for practice which are relevant to all to garner accurate sampling results. Particularly concerning is that 93.6% of respondents reported that positive proximal bone cultures would change their clinical management, yet only 64.5% of respondents would always take a proximal bone specimen. Evidence supporting the idea that culture from proximal bone specimens is of clinical benefit has been restricted only to small studies, and this may be because it is not part of local guidelines or protocols.10,11
The UK Standards for Microbiology Investigations acknowledges that surgery may be required in acute presentations of osteomyelitis and in chronic osteomyelitis where areas of dead bone may need resecting.9 Both need to be accompanied by specific antibiotic therapy depending on culture results. For bone, bone biopsies, soft tissues and aspirates, they recommend collecting specimens into appropriate CE marked leak-proof containers and putting them in sealed plastic bags with Ringer’s or saline solution and Ballotini beads (as an option) which is placed into sealed plastic bags. However, microbiology and histology specimen pots can be confused, leading to difficulties in processing samples. Specimens should be transported and processed as soon as possible. To enable timely clinical management, samples should be processed urgently. The Infectious Diseases Society of America guidelines recommend that specimens should be transported at room temperature and should be processed immediately, and within a maximum of 2 hours. If processing is delayed, refrigeration is preferable to storage at ambient temperature.12
The IWGDF advises on collecting an appropriate specimen for culture for almost all clinically infected wounds to determine the causative pathogens. For a soft tissue diabetic foot infection, a sample for culture should be obtained by aseptically collecting a tissue specimen (by curettage or biopsy) from the ulcer. During surgery to resect bone for diabetic foot osteomyelitis, obtaining a specimen of bone for culture (and if possible, histopathology) at the stump of the resected bone should be considered to identify if there is residual bone infection. If an aseptically collected culture specimen obtained during surgery grows pathogens or if the histology demonstrates osteomyelitis, appropriate antibiotics should be administered for up to 6 weeks.6
In our survey, 58.9% of responding consultants declared an interest in diabetes which remains concerning considering the epidemic proportions of the problem. This is a possible confounding factor within the results, as those with a specialist interest may be more engaged in pathway design and teaching sampling techniques. This should be considered when reviewing the results. Few stated that they had received training and the majority (62.1%) of these were only local departmental training. 66.4% of the respondents reported the absence of a formal local policy for diabetic foot sampling, leaving surgeons to draw on their own experience. This is of concern as it suggests that vascular surgeons, diabetic specialists (clinician and podiatrists) and microbiologists providing care may not all be engaged in the process of diabetic foot management using a coherent strategy.
Antibiotic stewardship remains a problem for the NHS and can only be guided by the results gained from sampling and the knowledge level of the clinicians involved in treatment decisions. With the plethora of techniques for sampling as described above, many of which have little or no proven benefit, clinicians must deliver care based on the results they have obtained and their own experience without the confidence that the samples are reproducible or accurate enough to guide antibiotic therapy.
Heterogenicity in training levels and teaching is presently being rectified by inclusion of the management of the diabetic foot within the core competency of the vascular training curriculum UK. It is imperative that trainees are given a solid grounding in diabetic foot management to build on when they enter consultant practice.7
Since the COVID pandemic the resources of the NHS are stretched and the ability to deliver care remains a problem. The long-term consequences of diabetic foot infection lead to a protracted hospital stay, multiple returns to theatre for surgery and the cost of major limb amputations with its hospital and social care costs.13 The recent NHS resolution document emphasises the importance of getting it right first time with diabetic foot infection as the costs of problems that develop are not inconsequential to the NHS and must be considered.1 Establishing good practice through training in diabetic foot infection and cooperation with the MDT can limit the requirement for intervention and reduce the burden on the NHS services.
Limitations
The survey achieved a 24% response rate from consultant members registered with VSGBI, providing a snapshot of opinions from an extensive geographical spread of UK vascular units. To help increase the response rate, an incentive was offered for completion and a reminder email was circulated at 4 and 8 weeks. Survey fatigue in general – and particularly at the end of the COVID 19 pandemic – cannot be ruled out as having an effect. This study is around an area of specific practice and therefore a level of inherent bias may have been introduced.
Conclusions
Practice for the sampling in diabetic foot infection by vascular surgeons remains varied across the country, with local policy guidelines only being present in certain locations. Lack of formal training is highly likely to be a significant contributing factor. A large proportion of vascular surgeons state that they have an interest in diabetic foot disease, and this must be drawn upon to standardise sampling techniques, develop treatment guidelines and deliver education and training going forward.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2023;2(4):200-204
Publication date:
July 17, 2023
Author Affiliations:
1. Black Country Vascular Network, Russell Hall Hospital, Dudley, UK
2. University Hospitals Birmingham NHS Foundation Trust, Birmingham, UK
3. Vascular Research and Innovation Consortium
4. University of Birmingham, Birmingham, UK
5. Birmingham City University, Birmingham, UK
Corresponding author:
Michael Lewis Wall
Black Country Vascular Network, Russell Hall Hospital,
Dudley, DY12HQ, UK
Email: [email protected]











