PROTOCOL
Protocol for the Physical Activity after Cardiovascular Screening (PACS) study in women aged 60–69 years: a prospective observational cohort study
Messeder SJ,1,2 Baldock SE,1 Rowlands AV,3 Barber S,4 Saratzis A,1,2 Bown MJ1,2
Plain English Summary
Why we are undertaking this work: Women are more likely than men to be misdiagnosed or receive poorer treatment for cardiovascular disease. Two key conditions – elevated blood pressure (BP) and peripheral artery disease (PAD) – are often missed in women. Both conditions are serious risk factors for heart disease and stroke. Screening and lifestyle advice, especially around exercise, can help reduce risk. This study was launched to find out if screening women for PAD and BP, and giving them tailored advice, could improve physical activity and health outcomes.
What we will do: We will invite women aged 60–69 years from Leicestershire to attend screening for PAD and high BP. Two hundred of these women will be invited to wear an activity tracker for 7 days at the time of screening and again 6 months later. One hundred women who are not being screened will be invited as a comparison. The goal is to track changes in physical activity levels and examine any lifestyle improvements after screening and receiving health advice.
What we expect to find: We expect to find that women who are diagnosed with PAD or high BP will increase their physical activity levels the most. This is compared with women who screen negative or who are not screened. We are also collecting information such as smoking and alcohol use and weight to see if the screening helps encourage broader health changes.
What this means: If our findings support the idea that screening leads to increased activity and healthier behaviours, it could justify rolling out targeted screening for women across the UK. This may help close the gender gap in cardiovascular care, promote healthier lifestyles and reduce the risk of heart attacks and strokes in women.
Abstract
Introduction: Women with cardiovascular disease receive a poorer standard of care than men; they are more likely to be misdiagnosed and undertreated. Individuals with peripheral artery disease (PAD) and/or elevated blood pressure (BP) have a similar cardiovascular risk as those with established ischaemic heart disease. As part of a National Institute for Health and Care Research (NIHR) programme, we are screening women for PAD and elevated BP. The primary objective is to determine key performance metrics for a screening programme: attendance and disease prevalence. There is, however, an opportunity to undertake a more detailed process evaluation of PAD+BP screening in women to help address this sex-based health inequality. We aim to assess whether screening and/or the identification of PAD/elevated BP, along with patient education in the form of leaflets and face-to-face advice, results in positive changes in physical activity.
Methods and analysis: The Physical Activity after Cardiovascular Screening (PACS) study is funded by the British Heart Foundation (FS/CRTF/23/24452) and is a sub-study of the NIHR PHAST-F study, an observational cross-sectional multicentre feasibility study (NIHR200601; UK’s Clinical Study Registry Registration ISRCTN17320335). Women in Leicestershire aged 60–69 years will be invited to attend for PAD+BP screening. Two hundred consenting participants (100 positive for PAD/or elevated BP and 100 negative) will undergo assessment of physical activity using accelerometers and complete the International Physical Activity Questionnaire Short Form, the EQ-5D-5L, Generalised Anxiety Disorder Questionnaire and the Edinburgh Claudication Questionnaire. Follow-up assessments will include accelerometers, questionnaires and GP data obtained at 6 months. An unscreened cohort of 100 women will also be recruited to evaluate the broader impact of screening.
Ethics and dissemination: The study has received ethical approval from the Sub-Committee of the North of Scotland Research Ethics Committee (reference 21/NS/0147). The results will be disseminated through research presentations, papers and social media.
Introduction
Women with cardiovascular disease receive a poorer standard of care than men; they are more likely to be misdiagnosed and undertreated.1 Contributing factors include failure to recognise the importance of secondary prevention, sex-related differences in clinical presentation, delays in seeking care and misperceptions about cardiovascular disease in women.1 These issues also contribute to elevated blood pressure (BP) and peripheral artery disease (PAD) being underdiagnosed and understudied in women.2
The pathophysiology of PAD is similar to coronary artery disease.3,4 Studies of asymptomatic individuals with PAD demonstrate a 5-year cardiovascular risk of around 20%.5,6 UK primary care data records the prevalence of diagnosed PAD at around 3%;7 however, large empirical population studies estimate the true prevalence to be between 13% and 18%.8-10 A lack of public awareness contributes to this under-diagnosis. In a recent small study of population screening for PAD, Davies et al.11 found that most individuals with PAD do not recognise their symptoms or present to health services.
Elevated BP is also often underdiagnosed due to its asymptomatic nature. It is estimated that up to 6–8 million adults within the UK could have undiagnosed or uncontrolled elevated BP.12 This is significant as it is one of the most important risk factors for cardiovascular disease and death. In fact, a linear relationship between BP and mortality from both ischaemic heart disease and stroke exists. Mortality from ischaemic heart disease and stroke doubles for every 20 mmHg systolic or 10 mmHg diastolic increase in BP.13
Physical activity is an important modifiable risk factor for both PAD and elevated BP, improving pain-free walking distance as well as overall cardiorespiratory fitness.2,14,15 Higher levels of physical activity are also associated with a lower risk of cardiovascular disease and reduction in all-cause mortality over the medium and long term.15 Exercise is a subset of physical activity that is planned, structured and repetitive with the aim of improving or maintaining physical fitness.16 In individuals with hypertension, exercise is as effective as antihypertensive medication in reducing systolic BP.17 A supervised exercise programme is the primary clinical management strategy for individuals with PAD.13,14,18 However, a recent audit19 showed that only 36% of UK vascular centres were able to offer supervised exercise therapy for PAD. Additionally, of those that did, only 6.8% were fully compliant with current National Institute for Health and Care Excellence (NICE) guidelines.14
Existing evidence
The Viborg vascular trial (VIVA),20 a Danish study that assessed the efficacy and cost-effectiveness of combined screening for PAD, BP and abdominal aortic aneurysm (AAA) in men aged 65–74 years, showed a significant reduction in all-cause mortality compared with no screening. The trial, however, did not assess objective physical activity levels and was unable to assess the impact that screening and clinical management had on physical exercise. A small population screening study from South Wales11 invited men aged 45 and women over 55 with cardiovascular risk factors for PAD screening. Similar to VIVA, this study did not assess the impact on physical activity levels.
Currently within the UK individuals aged 40–74 years with no pre-existing cardiovascular disease are invited for a free NHS Health Check every five years. During this appointment, individuals receive an assessment for BP, hypercholesterolaemia and body mass index as well as general cardiovascular advice focused on smoking cessation, physical activity and maintaining a healthy lifestyle. In 2023, only 40% of individuals invited for an NHS Health Check attended.21 In recent years there has been a trend towards lower rates of attendance, therefore more needs to be done to maximise cardiovascular health screening at every clinical encounter.
PHAST-F Study
The Peripheral arterial disease, High blood pressure and Aneurysm Screening Trial (PHAST) is a multicentre £2.4 million NIHR-funded programme examining the feasibility and effectiveness of screening men for PAD and elevated BP at the same time as screening for AAA (NIHR200601, Chief Investigator: Bown). As there is no AAA screening programme for women, the overall PHAST approach potentially exacerbates the sex-based inequality in preventative cardiovascular medicine highlighted above. To directly address this inequality, the PHAST programme includes a limited feasibility study of isolated BP and PAD screening for women (PHAST-F). This feasibility study is focused on determining attendance for screening and disease prevalence but is a good opportunity to undertake a more detailed evaluation of responses to PAD+BP screening in this population. The European Society for Vascular Surgery guidelines recommend the consideration of screening for PAD in individuals aged >70 years or 45–69 years with cardiovascular risk factors.22
The current National AAA Screening Programme invites men on the year of their 65th birthday. Therefore, women aged 60–69 years are invited to take part in this feasibility study to align closely with these two considerations.
Ongoing studies
A search of the clinicaltrials.gov, International Standard Randomised Controlled Trials Number and EU Clinical Trials registries identified no ongoing population screening trials in this area (PAD or elevated BP). We have not identified any ongoing trials of population screening for PAD or elevated BP funded by the NIHR or other UK institutions.
Why is this research needed now?
The PACS study supports the 10 Year Health Plan for England23 by preventing heart attacks and strokes, and aligns with the Department of Health and Social Care’s prioritisation24 for prevention medicine. In 2019 the National Cardiovascular Disease Prevention System Leadership Forum launched its cardiovascular disease prevention programme. Additionally, the Lancet has commissioned a global report on cardiovascular disease in women to tackle the inequality in cardiovascular health by 2030.25 This research is therefore timely as well as relevant. This study aims to assess if PAD+BP screening and providing women with information on disease pathology and treatment will ultimately increase physical activity levels and overall cardiovascular health.
Research hypothesis
Women who screen negative for PAD/elevated BP will have a greater level of physical activity at initial screening than women who screen positive. Women who screen positive for PAD/elevated BP will have a greater increase in physical activity levels from initial screening to 6 months than women who screen negative.
Outcomes
Primary outcome
Change in average daily milligravities (mg) of physical activity (measured using GENEActiv accelerometer (acceleration/steps)) and MVPA (defined as time accumulated above an average acceleration of 100 mg in bouts of at least 1 minute)26 from screening to 6 months.
Secondary outcomes
• Average daily mg of physical activity and moderate to vigorous physical activity (MVPA) in women aged 60–69 years
• Comparison of physical activity and MVPA at screening of women who screen negative for PAD/and or elevated BP and women who screen positive
• Comparison of average daily mg of physical activity and MVPA at screening for women who screen negative for PAD/and or elevated BP and women who screen positive
• Change in smoking, alcohol consumption, weight and subjectively documented physical activity levels in individuals who screen positive for elevated BP/and or PAD
• Time spent in 24 hours of physical behaviour intensities (inactive time, light physical activity, moderate physical activity and sleep) amongst all groups
Objectives
Primary objectives
To assess the average change in accelerometer-assessed physical activity/steps immediately after initial PAD+BP screening to 6 months in women.
Secondary objectives
• To determine the ‘normal’ physical activity levels of women aged 60–69 years
• To determine whether women who screen positive for elevated BP/PAD have lower baseline levels of physical activity than those who screen negative
• To determine whether women who screen positive for elevated BP/and or PAD have a greater increase in physical activity levels at 6 months than women who screen negative
• To determine whether elevated BP/and or PAD at screening results in a positive change in health behaviour
Methods and analysis
Study design
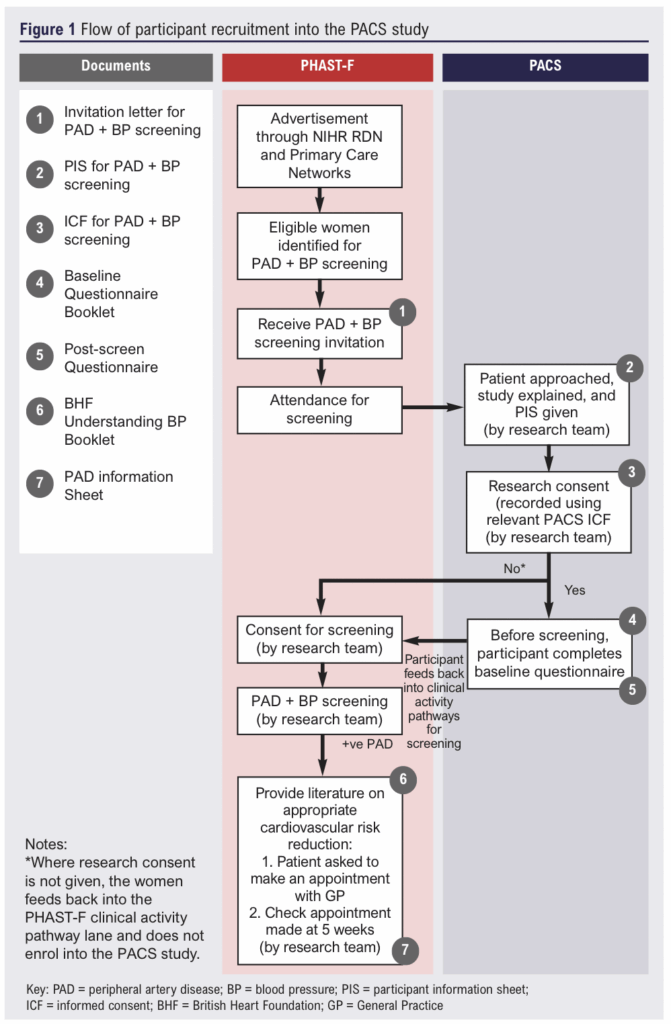
The Standard Protocol Items: Recommendations for Intervention Trials27 and the Strengthening the Reporting of Observational Studies in Epidemiology28 statements were followed in the development of this study. The Physical Activity after Cardiovascular Screening (PACS) study is a prospective observational cohort study to determine if screening for hypertension and PAD results in an increase in physical activity levels from baseline to 6 months after screening. PACS is funded by the British Heart Foundation (FS/CRTF/23/24452). It is a sub-study of the PHAST-F study, which is an observational cross-sectional multicentre feasibility study funded by the NIHR Programme Grants for Applied Research scheme (NIHR200601) (Figure 1).
Study setting
Women attending for PAD+BP screening as part of the PHAST-F study will be invited to participate in PACS. The NIHR Research Delivery Network will support the recruitment of Leicestershire general practices to take part in PHAST-F. In addition, individual general practices based in Leicestershire will be directly approached by members of the trial team.
Women aged 60–69 years registered with participating general practices will then be invited to attend for PAD+BP screening. Letters of invitation will be sent via post with patients asked to contact the department, either by telephone or email, to book a screening appointment at a convenient time for them. All letters will also include a PAD screening information leaflet (see Appendix 1 Supplementary file online at www.jvsgbi.com) and a local map.
Leicestershire has been chosen as the study location because it is a ‘plural city’ with no single ethnic majority. The wider Leicestershire area serves individuals from diverse socioeconomic and ethnic backgrounds across rural and urban areas.
Eligibility criteria
The target population for this study are women aged 60–69 years registered with a Leicestershire General Practice and attending the PHAST-F programme for PAD+BP screening. Patients will be eligible for the study if they fulfil the inclusion and exclusion criteria shown in Table 1.

Recruitment
Upon arrival at the screening clinic, women attending for PAD+BP screening will receive a participant information sheet (see Appendix 1 Supplementary file online at www.jvsgbi.com). Patients will be pre-screened by a member of the clinical team and approached to ask if they wish to speak to a member of the research team. It will be emphasised that declining participation in PACS will not affect their clinical care, and screening will proceed as planned. Patients who express interest will be directed to a member of the research team, who will confirm eligibility and obtain informed consent prior to screening.
Data collection
The following data will be collected at baseline and at 6 months:
• Demographics: age, sex, ethnicity, height, weight, smoking and alcohol use, occupation status (or previous occupation where retired)
• Cardiovascular history: hypertension, PAD, diabetes, hypercholesterolaemia, angina, myocardial infarction, stroke/transient ischaemia attack, cardiac arrhythmia, operation for coronary arteriosclerosis
• Medication history
• Patient-reported outcomes: European Quality of Life 5 Dimensions 5-level (EQ-5D-5L), Generalised Anxiety Disorder Questionnaire (GAD-7), Edinburgh Claudication Questionnaire (ECQ) and International Physical Activity Questionnaire Short Form (IPAQ-SF)
• Health utilisation (for health economics analysis): Accident and Emergency attendance, hospital admissions, hospital outpatient appointments, primary care services accessed
Intervention: PAD+BP screening
A systematic review of the diagnostic accuracy of automated and semi-automated ankle-brachial pressure index (ABPI)/toe pressure devices and a consensus conference to determine real-world usability was previously undertaken to determine the most suitable device for PHAST-F.29 Trained healthcare professionals will perform the ABPI using the MESI-ABPI-MD device.30 Measurements will be obtained after a period of rest in the supine position. Systolic BP will be measured simultaneously in one arm (brachial artery) and both ankles (posterior tibial arteries). The test is then repeated using the alternate arm. The ABPI is automatically calculated by the MESI-ABPI-MD device and the reading will be verified by healthcare professionals. The ABPI reading will be the lowest of the two readings. All results obtained during screening will be sent to the patients’ general practitioner.
Elevated BP will be defined as either a systolic BP of >140 mmHg or a diastolic BP of >90 mmHg and individuals will receive a British Heart Foundation (BHF) ‘Understanding Blood Pressure’ booklet.31 The BHF booklet provides advice on physical activity with suggested guidance on how to increase levels. PAD will be defined as an ABPI of <0.9, as per NICE Guidelines.14 Individuals diagnosed with PAD will receive a PAD Information Sheet (see Appendix 1 Supplementary file online at www.jvsgbi.com). The PAD Information Sheet also provides advice on physical activity. All patients diagnosed with elevated BP and/or PAD will be asked to make an appointment with their GP to discuss their results further and undergo further investigations, if required. For all individuals who screen positive, the research team will make a follow-up telephone call appointment at 5 weeks to discuss what health behaviour changes (cessation of smoking, reduction in alcohol consumption, maintenance of a healthy weight and increase in subjectively documented physical activity levels), if any, have been adopted.
Follow-up
Follow-up will be conducted remotely and will take place at 6 months. The data collection mentioned above will be collected via post. Participants who do not return 6-month GENEActiv results will be contacted by telephone. GP data and routinely collected data will also be obtained throughout the follow-up period. Data collected will include: date of GP appointment, BP at GP attendance, result of home BP diary, changes in medication, diagnosis of other cardiovascular-related risk factors (hypercholesterolaemia, stroke/transient ischaemic attack, angina, myocardial infarction, diabetes, major limb amputation, intervention for PAD), referral to secondary care services and associated outcomes. These data will be obtained to examine uptake and adherence to cardiovascular risk management following self-made GP appointments for those who screen positive for PAD/and or elevated BP.
Accelerometer
The accelerometer used will be the GENEActiv device. This device has been used in a wide range of clinical trials and is a well-recognised validated device for research in physical activity. Participants will provide consent to wear a GENEActiv accelerometer on their non-dominant wrist (defined as their non-writing wrist) 24 hours a day for 7 days. The GENEActiv will be initialised to begin recording at 00:01 hours at 100 Hz. Participants will be encouraged to go to bed wearing the device the evening of their screening appointment. They will also complete a self-reported diary stating the time they went to bed, estimated time they went to sleep, when they woke up in the morning and when they physically got out of bed. These results will be analysed in conjunction with the accelerometer results. Follow-up GENEActiv devices will be sent out to participants at 6 months via postal services along with the above questionnaires.
Unscreened group
We recognise that recruiting women for PACS who are attending for PAD+BP screening may influence baseline physical activity levels. We will invite women from the same population and age range as those invited for PAD+BP screening. To facilitate this, participants will be recruited through the Extended Cohort for E-health, Environment, and DNA (EXCEED) study. The EXCEED protocol has been described previously.32 Briefly, EXCEED is a longitudinal health study assessing the impact of genetics and lifestyle on long-term health conditions. The EXCEED study team were approached to help with recruitment into the unscreened ‘control’ group. A data access proposal form was approved by the core group in EXCEED with changes approved by the Sub-Committee of the North of Scotland Research Ethics Committee (Reference 21/NS/0147). Participants enrolled in the EXCEED study who have agreed to be contacted for future research will be invited to participate in PACS. Email invitations will be sent out from the EXCEED team until 100 women aged 60–69 years living in Leicestershire have enrolled into the PACS study. Consent will be obtained using one of the following methods, depending on participant preference and logistical feasibility: e-signature, email response, email attachment, postal consent, verbal consent (documented by the researcher), or written in-person consent. The unscreened group will wear a GENEActiv accelerometer and complete questionnaires, as described above.
Statistical analysis and plan
Sample size
We know that an increase of 1000 steps or 10 minutes of brisk walking corresponds to an increase of roughly 2 mg of average acceleration.33 Previous data regarding accelerometers corresponds to initial activity levels rather than degree of change. Therefore, our power calculation is based on the outcome of mg at 6 months. A sample size of 200 participants (100 positive for PAD/or elevated BP and 100 negative) would provide >80% power to detect a difference of 4 mg (roughly 2000 steps or 20 minutes of brisk walking). This would be at the 5% significance level, providing more than 61% of participants reached the 6-month time point (based on a standard deviation of ±7.8 mg). PAD and elevated BP outcomes will be analysed together as exercise is the primary treatment for both cardiovascular diseases. We will also undertake a sub-group analysis between women who are positive for PAD and women who are positive for elevated BP.
Unscreened group
We will aim for an attendance of 100 participants as a baseline convenience sample to compare physical activity levels of women who undergo PAD+BP screening with those who do not receive PAD+BP screening.
Accelerometer processing
The GENEActiv data will be uploaded using GENEActiv PC software version 3.3 and analysed using the latest version of the R-package GGIR version in R (http://cran.r-project.org). The approach for GENEActiv analysis has been described previously.34-37 Briefly, local gravity will be used as a reference for autocalibration,35 sustained abnormally high values will be detected and the average magnitude of dynamic acceleration will be calculated (corrected for gravity and expressed as Euclidean Norm Minus One (ENMO) in mg averaged over 5 s epochs). Participants will be excluded if their accelerometer files show a post-calibration error of >0.01 g (10 mg), <3 days of valid wear (defined as >16 hours per day), or wear data are not present for each 15 min period of the 24 hour cycle.36 The default non-wear setting in GGIR will be used, which imputes invalid data by the average at similar timepoints on different days of the week.36
The following measures will be generated and averaged across all valid days:
1. Average acceleration – used as a proxy measure of overall volume of physical activity (24-hour day). Higher levels of average acceleration correspond to higher levels of physical activity. The minimum clinically important difference is an increase in average acceleration by 1 mg a day38 (equivalent to a 5-minute brisk walk; 15-minute slow walk; or 500 daily steps).33
2. The intensity gradient – used to describe the distribution of physical activity intensity across the day.37 The intensity gradient is helpful to describe an individual’s activity profile. The steeper the gradient, the less time spent at higher levels of acceleration. Thus, the intensity gradient is always negative.
3. MVPA – defined as the time accumulated above an average acceleration of 100 mg in bouts of at least 1 minute (ie, moderate physical activity as described below).26
4. Time spent in different physical behaviour intensities across 24 hours:39
• Inactive time (defined as time accumulated below 40 mg)39
• Light physical activity (defined as time accumulated 40–99 mg)
• Moderate physical activity (defined as time accumulated 100–399 mg)
• Vigorous physical activity (defined as time accumulated >400 mg)
Data analysis
Categorical data will be presented as absolute values and proportions (%) with a χ2 test used to compare proportions between groups. Where data are paired, McNemar’s test will be used (ie, baseline to 6 months) or Fisher’s exact test if the sample size is less than five. Where more than two categories are present (never smoker; previous smoker; current smoker), then a χ2 test will be used.
Continuous variables will be examined for normality using histograms, skewness and kurtosis. Normally distributed variables will be reported as mean (± standard deviation) with a t-test (independent, ie, screen negative vs screen positive group) or a paired t-test (paired, ie, baseline to 6 months) used to compare within or between groups. For non-normally distributed variables, median (interquartile range) with a Wilcoxon rank-sum test (independent) or a Wilcoxon signed-rank test (paired) will be used to compare differences between groups. Where comparisons are undertaken across more than two groups (ie, control vs screen negative vs screen positive), ANOVA (normally distributed) or a Kruskal–Wallis test (non-normally distributed) will be used.
All analyses will be performed using R Core Team (Version 4.4.1; 2024; R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/).
Data management
Data will be recorded directly into the paper case report form (source data) and then transcribed into the electronic case report form. Original copies of the participant consent form and information sheet will be filed in the Investigator Site with copies placed in the participant’s hospital notes. Data entry will be conducted by the site research team and the Leicester Clinical Trials Unit (LCTU). Participant contact details will be securely collected and retained to enable send out of follow-up questionnaires by post. Data management will be through MACRO, a validated web-based data entry system and database.
Study management
The management of PACS will be undertaken by SJM, a British Heart Foundation Clinical Training Fellow. The management of PHAST-F will be undertaken by the LCTU, a fully registered UK Clinical Research Collaboration Clinical Trials Unit (UKCRC no.43) in collaboration with the Investigators and the Trial Management Group. The LCTU will be responsible for site set-up, project management, statistical expertise and provision of MACRO databases. Serious adverse events are not expected. The study intervention consists of PAD and BP screening, which are known to be safe with no long-term physical consequences.
Dissemination
We will disseminate and present our results at national UK conferences such as the Vascular Societies’ Annual Scientific Meeting. The results of this will be published in a peer-reviewed academic journal as it will be relevant for other researchers in cardiovascular medicine, psychology, epidemiology, public health and vascular surgery. It will inform these future researchers if physical activity levels could be used as a measurable outcome in screening. It will also inform the design of our future NIHR Health Technology Assessment Programme to see if physical activity monitoring can be used as an outcome for cardiovascular screening.
We will also publish outputs on social media in lay format and use existing PAD and diabetes patient groups to involve patients in dissemination, as well as our PPI group. This will improve patient education and ultimately cardiovascular health. For those involved in PACS, it represents an opportunity to discuss cardiovascular risk factor modification. The encouragement of a healthy lifestyle and exercise will lead to improved cardiovascular risk management and be of direct benefit to patients.

Article DOI:
Journal Reference:
J.Vasc.Soc.G.B.Irel. 2025;5(1):15-22
Publication date:
November 24, 2025
Author Affiliations:
1. Department of Cardiovascular Sciences, NIHR Leicester Biomedical Research Centre, University of Leicester, Glenfield Hospital, Leicester, UK
2. Leicester Vascular Institute, University Hospitals of Leicester, Glenfield Hospital, Leicester, UK
3. Leicester Diabetes Centre, University of Leicester, Leicester, UK
4. Leicester Clinical Trials Unit, University of Leicester, Leicester, UK
Corresponding author:
Dr Sarah Jane Messeder
Department of Cardiovascular Sciences, University of Leicester, Leicester LE3 9QP, UK
Email: [email protected]











